
Concept explainers
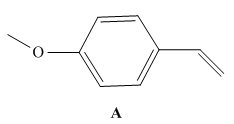
As we will learn in Chapter 30, styrene derivatives such as A can be polymerized by way of cationic rather than radical intermediates. Cationic
a. Draw a short segment of the
b. Why does A react faster than styrene
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
- What monomer is needed to form each polymer?arrow_forward. What do the prefixes ortho-, meta-, and para- refer to in terms of the relative location of substituents in a disubstituted benzene?arrow_forwardExplain why styrene (CH2=CHPh) can be polymerized to polystyrene by all three methods of chain-growth polymerization.arrow_forward

 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning





