
Concept explainers
Interpretation:
The energy required to heat a given amount of ethanol is to be calculated providing specific heat of ethanol.
Concept introduction:
Specific heat is defined as quantity of heat required to raise the temperature of one gram substance by one Kelvin. It is represented by ‘c’.
Answer to Problem 15STP
A
Explanation of Solution
The quantity of heat required can be calculated by using formula:
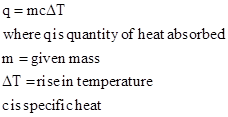
Given data:
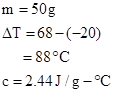
Substituting the values in formula:
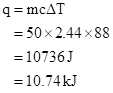
Thus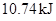 heat is required to raise the temperature of given amount of ethanol.
heat is required to raise the temperature of given amount of ethanol.
Chapter 15 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
CHEMISTRY-TEXT
Chemistry: The Central Science (14th Edition)
Organic Chemistry (8th Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
Chemistry: Structure and Properties (2nd Edition)
Chemistry: Structure and Properties
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





