
Concept explainers
Interpretation:
The value of 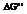 is to be calculated for the given reactions using the free-energy values from the
is to be calculated for the given reactions using the free-energy values from the 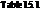 .
.
Concept introduction:
Gibbs free energy is the standard free energy that complements the production of  mole of that substance from its component elements, at their standard states.
mole of that substance from its component elements, at their standard states.
The symbol of standard Gibbs free energy is 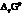 .
.
The mitochondrion is called the powerhouse of the cell.
Mitochondria is used for the synthesis of 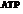 that is used to store the energy
that is used to store the energy
Answer to Problem 16RE
Solution:
The reaction will not proceed in the direction as stated. The value of 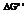 is
is 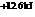 .
.
The reaction will proceed in the direction as stated. The value of 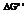 is
is 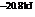 .
.
The reaction will not proceed in the direction as stated. The value of 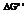 is
is 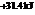 .
.
The reaction will proceed in the direction as stated. The value of 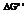 is
is 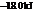 .
.
Explanation of Solution
Given information:
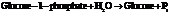
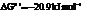
a) For predicting the reaction and calculating the value of 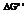 :
:
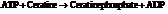
From the table 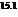 in the book,
in the book,
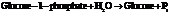
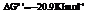
Also,
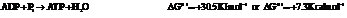
On reversing the second reaction,
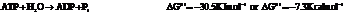
Now add the two reactions and their Gibbs free energies as follows:
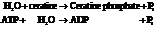
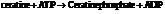
Substitute the value as follows:
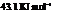 for
for 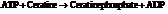 and
and
for
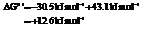
Hence, the reaction will not be in the direction as written.
b) For predicting the reaction and calculating the value of 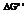 :
:
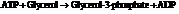
From the table 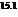 in the book,
in the book,
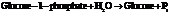
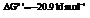
Also,
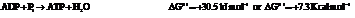
On reversing the second reaction,
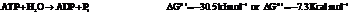
Now add the two reactions and their Gibbs free energies as follows:
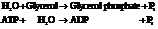
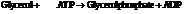
Substitute the value as follows:
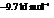 for
for 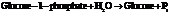 and
and
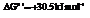 for
for 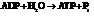
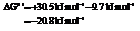
Hence, the reaction will be in the direction as written.
c) For predicting the reaction and calculating the value of 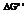 :
:
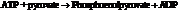
From the table 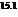 in the book,
in the book,
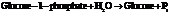
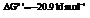
Also,
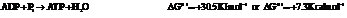
On reversing the second reaction,
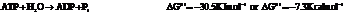
Now add the two reactions and their Gibbs free energies as follows:
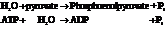
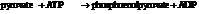
Substitute,
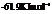 For
For 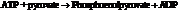 and
and
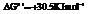 For
For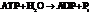 .
.
So,
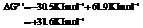
Hence, the reaction will not be in the direction as written.
d) For predicting the reaction and calculating the value of 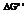 :
:
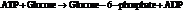
From the table 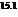 in the book,
in the book,
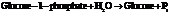
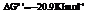
Also,
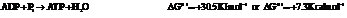
On reversing the second reaction,
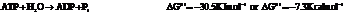
Now add the two reactions and their Gibbs free energies as follows:
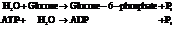

Substitute the value as follows:
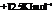 for
for 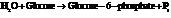 and
and
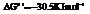 for.
for. 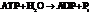
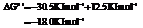
Hence, the reaction will be in the direction as written.
Want to see more full solutions like this?
Chapter 15 Solutions
Biochemistry
- REFLECT AND APPLY Two biochemistry students are about to use mitochondria isolated from rat liver for an experiment on oxidative phosphorylation. The directions for the experiment specify addition of purified cytochrome c from any source to the reaction mixture. Why is the added cytochrome c needed? Why does the source not have to be the same as that of the mitochondria?arrow_forwardREFLECT AND APPLLY What is the metabolic advantage of having both hexokinase and glucokinase to phosphorylate glucose?arrow_forwardREFLECT AND APPLLY Is the reaction of 2-phosphoglycerate to phosphoenolpyruvate a redox reaction? Give the reason for your answer.arrow_forward
- REFLECT AND APPLY A model is proposed to explain the reaction catalyzed by an enzyme. Experimentally obtained rate data fit the model to within experimental error. Do these findings prove the model?arrow_forwardREFLECT AND APPLY What is the relationship between a transition-state analog and the induced-fit model of enzyme kinetics?arrow_forwardMATHEMATICAL Using the information in Table 20.2, calculate G for the following reaction: 2Cytaa3[oxidized;Fe(III)]+2Cytb[reduced;Fe(II)]2Cytaa3[reduced;Fe(II)]+2Cytb[oxidized;Fe(III)]arrow_forward
- REFLECT AND APPLY What do the following indicators tell you about whether a reaction can proceed as written? (a) The standard free-energy change is positive. (b) The free-energy change is positive. (c) The reaction is exergonic.arrow_forwardREFLECT AND APPLLY What are the metabolic effects of not being able to produce the M subunit of phosphofructokinase?arrow_forwardMATHEMATICAL Consider the reaction AB+C, where G=0.00. (a) What is the value of G (not G) when the initial concentrations of A, B, and C are 1 M, 103M,and106M? (b) Try the same calculations for the reaction D+EF, for the same relative order of concentrations. (c) Try the same calculations for the reaction GH, if the concentrations are 1Mand103M for G and H, respectively.arrow_forward
- REFLECT AND APPLY In metabolism, glucose-6-phosphate (G6P) can be used for glycogen synthesis or for glycolysis, among other fates. What does it cost, in terms of ATP equivalents, to store G6P as glycogen, rather than to use it for energy in glycolysis? Hint: The branched structure of glycogen leads to 90% of glucose residues being released as glucose-1-phosphate and 10% as glucose.arrow_forwardREFLECT AND APPLY Metabolic cycles are rather common (Calvin cycle, citric acid cycle, urea cycle). Why are cycles so useful to organisms?arrow_forwardMATHEMATICAL For an enzyme that displays MichaelisMenten kinetics, what is the reaction velocity, V (as a percentage of Vmax), observed at the following values? (a) [S]=KM (b) [S]=0.5KM (c) [S]=0.1KM (d) [S]=2KM (e) [S]=10KMarrow_forward
 BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
