
Concept explainers
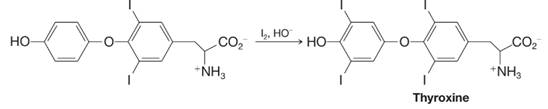
The structure of thyroxine, a thyroid hormone that helps to regulate
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
Additional Science Textbook Solutions
Introductory Chemistry (5th Edition) (Standalone Book)
General, Organic, and Biological Chemistry (3rd Edition)
Chemistry: Structure and Properties (2nd Edition)
Chemistry
Chemistry: A Molecular Approach
Introductory Chemistry (6th Edition)
- Please explain the synthesis. Identify SN1, SN2, E1, E2, nucleophiles and ekectrophiles.arrow_forwardArrange the compounds in order of INCREASING reactivity towards bromination. Toluene, Nitrobenzene, Anisole, Aniline Acetophenone, Bromobenzene, Aniline, Phenol Acetanilide, Benzaldehyde, Toluene, Iodobenzenearrow_forwardIbufenac, a para-disubstituted arene with the structure HO2CCH2C6H4CH2CH(CH3)2, is a much more potent analgesic than aspirin, but it was never sold commercially because it caused liver toxicity in some clinical trials. Devise a synthesis of ibufenac from benzene and organic halides having fewer than five carbons.arrow_forward
- Isoamyl acetate (also known as isopentyl acetate) is an ester that is referred to as “banana flavor” due to its odor that resembles that of banana. It can be synthesized from isopentyl alcohol and acetic acid via nucleophilic acyl substitution mechanism. Propose a mechanism for the synthesis of isoamyl acetate from acetic acid and isoamyl alcohol.arrow_forwardRank the following carboxylic acid derivatives in decreasing order (most to least) of reactivity towards nucleophilic acyl substitution.arrow_forwardThe compound eutypine is an antibacterial agent isolated from the fungus Eutypa lata. This fungus results in a disease common to vineyards called eutyposis. Give a sequence of reactions that will take the following reactant and give eutypine when the other reactants used in the sequence are acetylene and acetone.arrow_forward
- Propose a mechanism for the following reaction, an important step in the laboratory synthesis of proteins:arrow_forwardAcid-catalyzed hydrolysis of the following epoxide gives a trans diol. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT



