
Concept explainers
Propose structures for
(a) C9H12; gives only one C9H11Br product on substitution of a hydrogen on the aromatic ring with bromine
(b) C10H14; gives only one C10H13Cl product on substitution of a hydrogen on the aromatic ring with chlorine
(c) C8H10; gives three C8H9Br products on substitution of a hydrogen on the aromatic ring with bromine
(d) C10H14; gives two C10H13Cl products on substitution of a hydrogen on the aromatic ring with chlorine
a) C9H12; gives only one C9H11Br product on substitution of a hydrogen on the aromatic ring with bromine
Interpretation:
A possible structure for the hydrocarbon with molecular formula C9H12 that gives only one monobromination product C9H11Br on substitution of a hydrogen on the aromatic ring with bromine is to be given.
Concept introduction:
All aromatic compounds are derivatives of benzene. The benzene ring accounts for six carbon atoms. The remaining carbons can be attached as alkyl group like methyl, ethyl etc. to the benzene ring.
To propose:
A possible structure for the hydrocarbon with molecular formula C9H12 that gives only one monobromination product C9H11Br on substitution of a hydrogen on the aromatic ring with bromine.
Answer to Problem 23AP
A possible structure for the hydrocarbon with molecular formula C9H12 that gives only one monobromination product C9H11Br on substitution of a hydrogen atom on the aromatic ring with bromine is
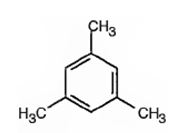
Explanation of Solution
The molecular formula of the compound is C9H12. Benzene ring accounts for six carbons. When the three carbons remaining are arranged as three methyl groups alternatively on the benzene ring the three hydrogens on the other three carbons become equivalent. Hence upon bromination, the hydrocarbon gives only one monosubstituted product.
A possible structure for the hydrocarbon with molecular formula C9H12 that gives only one monobromination product C9H11Br on substitution of a hydrogen atom on the aromatic ring with bromine is

b) C10H14; gives only one C10H13Cl product on substitution of a hydrogen on the aromatic ring with chlorine
Interpretation:
A possible structure for the hydrocarbon with molecular formula C10H14 that gives only one monochlorination product C10H13Cl on substitution of a hydrogen on the aromatic ring with chlorine is to be given.
Concept introduction:
All aromatic compounds are derivatives of benzene. The benzene ring accounts for six carbon atoms. The remaining carbons can be attached as alkyl group like methyl, ethyl etc. to the benzene ring. Only one monosubstituted product will be obtained only if all the hydrogen atoms in the aromatic ring are equivalent.
To propose:
A possible structure for the hydrocarbon with molecular formula C10H14 that gives only one monochlorination product C10H13Cl on substitution of a hydrogen on the aromatic ring with chlorine is to be given.
Answer to Problem 23AP
A possible structure for the hydrocarbon with molecular formula C10H14 that gives only one monochlorination product, C10H13Cl, on substitution of a hydrogen atom on the aromatic ring with chlorine is
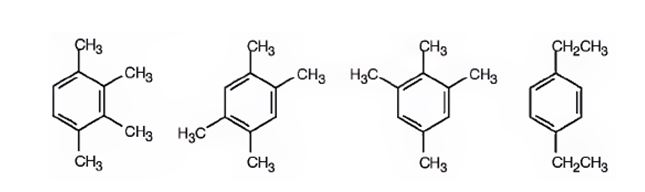
Explanation of Solution
The molecular formula of the hydrocarbon is C10H14. Benzene ring accounts for six carbons. The remaining four carbons can be arranged as four methyl groups or as two ethyl groups on the benzene ring in four different ways as shown such that remaining hydrogen atoms become equivalent. Hence upon chlorination, the hydrocarbon will give only one monosubstituted product.
A possible structure for the hydrocarbon with molecular formula C10H14 that gives only one monochlorination product, C10H14Cl, on substitution of a hydrogen atom on the aromatic ring with chlorine is

c) C8H10; gives three C8H9Br products on substitution of a hydrogen on the aromatic ring with bromine
Interpretation:
A possible structure for the hydrocarbon with molecular formula C8H10 that gives three monobromination products. C8H9Br, on substitution of a hydrogen on the aromatic ring with bromine is to be given.
Concept introduction:
All aromatic compounds are derivatives of benzene. The benzene ring accounts for six carbon atoms. The remaining carbons can be attached as alkyl group like methyl, ethyl etc. to the benzene ring.
To propose:
A possible structure for the hydrocarbon with molecular formula C8H10 that gives three products, C8H9Br, on substitution of a hydrogen on the aromatic ring with bromine.
Answer to Problem 23AP
A possible structure for the hydrocarbon with molecular formula C8H10 that gives three monobromination products C9H11Br on substitution of a hydrogen atom on the aromatic ring with bromine is
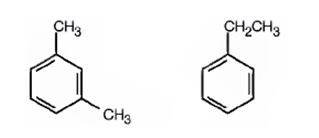
Explanation of Solution
The molecular formula of the hydrocarbon is C8H10. Benzene ring accounts for six carbons. When the two carbons remaining are arranged as two methyl groups meta to each other or as an ethyl group on the benzene ring hydrogens on the other carbon atoms in the ring classify themselves into three different groups. Hence upon bromination, the hydrocarbon gives three monosubstituted products.
A possible structure for the hydrocarbon with molecular formula C8H10 that gives three monobromination products, C9H11Br, on substitution of a hydrogen atom on the aromatic ring with bromine is

d) C10H14; gives two C10H13Cl products on substitution of a hydrogen atom on the aromatic ring with chlorine
Interpretation:
A possible structure for the hydrocarbon with molecular formula C10H14 that gives two monochlorination products C10H13Cl on substitution of a hydrogen atom on the aromatic ring with chlorine is to be given.
Concept introduction:
All aromatic compounds are derivatives of benzene. The benzene ring accounts for six carbon atoms. The remaining carbons can be attached as alkyl group like methyl, ethyl etc. to the benzene ring.
To propose:
A possible structure for the hydrocarbon with molecular formula C10H14 that gives two products, C10H13Cl, on substitution of a hydrogen on the aromatic ring with chlorine.
Answer to Problem 23AP
Possible structure for the hydrocarbon with molecular formula C10H10 that gives two monobromination products, C10H13Cl, on substitution of a hydrogen atom on the aromatic ring with chlorine is
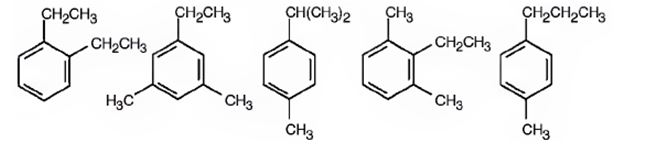
Explanation of Solution
The molecular formula of the compound is C10H14. Benzene ring accounts for six carbons. The four carbons remaining can be arranged as two ethyl groups or as an ethyl & two methyl groups or as propyl & methyl groups or as isopropyl & methyl groups on the benzene ring as shown so that the hydrogens on the other carbon atoms in the ring classify themselves into two different groups. Hence upon chlorination, the hydrocarbon gives two monosubstituted products.
A possible structure for the hydrocarbon with molecular formula C10H10 that gives two monobromination products, C10H13Cl, on substitution of a hydrogen atom on the aromatic ring with chlorine is

Want to see more full solutions like this?
Chapter 15 Solutions
Organic Chemistry - Owlv2 Access (4 Term)
- Aromatic substitution can be done on naphthalene. Treatment of naphthalene with concentrated H2SO4 gives two (and only two) different sulfonic acids.arrow_forwardDeduce the structure of each compound from the information given. All unknowns in this problem have molecularformula C8H12.(a) Upon catalytic hydrogenation, unknown W gives cyclooctane. Ozonolysis of W, followed by reduction with dimethylsulfide, gives octanedioic acid, HOOC¬(CH2)6¬COOH. Draw the structure of W.(b) Upon catalytic hydrogenation, unknown X gives cyclooctane. Ozonolysis of X, followed by reduction with dimethylsulfide, gives two equivalents of butanedial, O“CH¬CH2CH2¬CH“O. Draw the structure of X.(c) Upon catalytic hydrogenation, unknown Y gives cyclooctane. Ozonolysis of Y, followed by reduction with dimethylsulfide, gives a three-carbon dialdehyde and a five-carbon dialdehyde. Draw the structure of Y.arrow_forwardDeduce the structure of each compound from the information given. All unknowns in this problem have molecularformula C8H12.(a) Upon catalytic hydrogenation, unknown W gives cyclooctane. Ozonolysis of W, followed by reduction with dimethylsulfide, gives octanedioic acid, HOOC¬(CH2)6¬COOH. Draw the structure of W.(b) Upon catalytic hydrogenation, unknown X gives cyclooctane. Ozonolysis of X, followed by reduction with dimethylsulfide, gives two equivalents of butanedial, O“CH¬CH2CH2¬CH“O. Draw the structure of X.(c) Upon catalytic hydrogenation, unknown Y gives cyclooctane. Ozonolysis of Y, followed by reduction with dimethylsulfide, gives a three-carbon dialdehyde and a five-carbon dialdehyde. Draw the structure of Y.*(d) Upon catalytic hydrogenation, unknown Z gives cis-bicyclo[4.2.0]octane. Ozonolysis of Z, followed by reductionwith dimethyl sulfide, gives a cyclobutane with a three-carbon aldehyde (¬CH2¬CH2¬CHO) group on C1 and aone-carbon aldehyde (¬CHO) group on C2. Draw the…arrow_forward
- Compound A (C7H11Br) is treated with magnesium in ether to give B (C7H11MgBr) which reacts violently with D2O to give 1-methylcyclohexene with a deuterium atom on the methyl group (C). Reaction of B with acetone (Ch3COCH3) followed by hydrolysis gives D (C10H18O). Heating D with concentrated H2SO4 gives E (C10H16), which reacts with 2 equivalents of Br2 to give F (C10H16Br4). E undergoes hydrogenation with excess H2 and Pt catalyst to give 2-methylpropylcyclohexane. Determine the structures of compounds A through F, and show your reasoning throughout.arrow_forwardThe compound MON-0585 is a non-toxic, biodegradable larvicide that is highly selective against mosquito larvae. Synthesize MON-0585 using either benzene or phenol as a source of the aromatic rings.arrow_forwardA certain hydrocarbon, C5H6, yields the two products shown below upon ozonolysis with 1. O3/CH2Cl2 2. Zn/H3O . Deduce the structure of the hydrocarbon.arrow_forward
- Starting with benzene, toluene, or phenol as the only sources of aromatic rings, show how to synthesize the following. Assume in all syntheses that mixtures of ortho-para products can be separated into the desired isomer. Q.)2,4,6-Trinitrotoluene (TNT)arrow_forwardPropose the structure of the following: a. An alkane, C6H14 b. A crylic saturated hydrocarbon, C6H12 c. A diene (dialkene), C5H8 d. A keto alkene, C5H8Oarrow_forwardThe following reaction was performed as part of a research program sponsored by the National Institutes of Health to develop therapeutic agents for the treatment of cocaine addiction. Using what you have seen about the reactions of halogens with alkenes, propose a mechanism for this process.arrow_forward
- Treatment of (CH3)2CHCH(OH)CH2CH3 with TsOH affords two products(M and N) with molecular formula C6H12. The 1H NMR spectra of M and Nare given below. Propose structures for M and N, and draw a mechanismto explain their formation.arrow_forward1. Deduce the structure of each compound from the information given. All unknowns in this problem have molecular formula C8H12. (a) Upon catalytic hydrogenation, unknown W gives cyclooctane. Ozonolysis of W, followed by reduction with dimethylsulfide, gives octanedioic acid, HOOC—(CH2)6—COOH. Draw the structure of W. (b) Upon catalytic hydrogenation, unknown X gives cyclooctane. Ozonolysis of X, followed by reduction with dimethyl sulfide, gives two equivalents of butanedial, O=CH—CH2CH2—CH=O. Draw the structure of X.arrow_forwardCompound A (C7H11Br) is treated with magnesium in ether to give B (C7H11MgBr), which reacts violently with D2O to give 1-methylcyclohexene with a deuterium atom on the methyl group (C). Reaction of B with acetone (CH3COCH3) followed by hydrolysis gives D (C10H18O). Heating D with concentrated H2 SO4 gives E (C10 H16), which decolorizes two equivalents of Br2 to give F (C10H16 Br4). E undergoes hydrogenation with excess H2 and a Pt catalyst to give isobutylcyclohexane. Determine the structures of compounds A through F, and show your reasoning throughout.arrow_forward
