
(a)
Interpretation: The electron dot structures of
Concept introduction: Electron dot structures or Lewis structures represents the bonding present between two atoms of a molecule and also shows the lone pair of electrons present in a particular element.
(a)
Answer to Problem 112A
The electron dot structures of
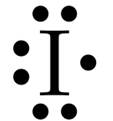
Explanation of Solution
The electronic structure of elements is represented with the help of an electron dot structure. The number of valence electrons present in each element is represented with the help of dots.
For iodine with the
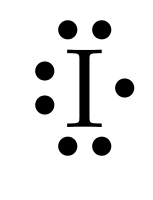
(b)
Interpretation: The electron dot structures of
Concept introduction: Electron dot structures or Lewis structures represents the bonding present between two atoms of a molecule and also shows the lone pair of electrons present in a particular element.
(b)
Answer to Problem 112A
The electron dot structures of

Explanation of Solution
For tellurium with the atomic number 52, the valence electrons are six. So the Lewis dot structure involves six dots surrounding the symbol of the element,

(c)
Interpretation: The electron dot structures of
Concept introduction: Electron dot structures or Lewis structures represents the bonding present between two atoms of a molecule and also shows the lone pair of electrons present in a particular element.
(c)
Answer to Problem 112A
The electron dot structures of
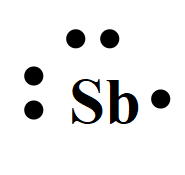
Explanation of Solution
For antimony with the atomic number 51, the valence electrons are five. So the Lewis dot structure involves five dots surrounding the symbol of the element,
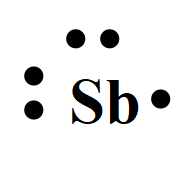
(d)
Interpretation: The electron dot structures of
Concept introduction: Electron dot structures or Lewis structures represents the bonding present between two atoms of a molecule and also shows the lone pair of electrons present in a particular element.
(d)
Answer to Problem 112A
The electron dot structures of
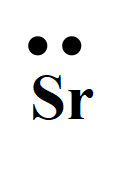
Explanation of Solution
For strontium with the atomic number 38, the valence electron is two. So, the Lewis dot structure involves two dots surrounding the symbol of the element,
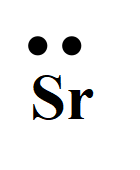
Chapter 16 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





