
Concept explainers
(a)
Interpretation:
The structure of the
Concept Introduction:
An aldehyde refers to a chemical compound which contains -CHO functional group that consists of a carbonyl center (carbon -oxygen double bond) provided that the carbon atom is attached to hydrogen and R group of the carbon chain. A molecular formula depicts the
Answer to Problem 15P
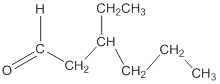
Explanation of Solution
To draw the structure of an aldehyde having the molecular formula
Thus, the structure of the aldehyde can be represented as follows:
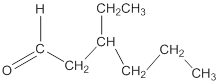
(b)
Interpretation:
The structure of the
Concept Introduction:
In chemistry a ketone functional group refers to the structure with general molecular formula
Answer to Problem 15P
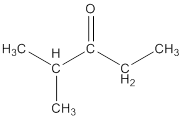
Explanation of Solution
To draw the structure of ketone having molecular formula
The structure so formed is shown below:
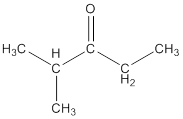
(c)
Interpretation:
The structure of ketone having the molecular formula
Concept Introduction:
In chemistry a ketone functional group refers to the structure with general molecular formula
Answer to Problem 15P
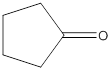
Explanation of Solution
To draw the structure of ketone having the molecular formula
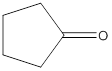
(d)
Interpretation:
The structure of an aldehyde with the molecular formula
Concept Introduction:
An aldehyde refers to a chemical compound which contains -CHO functional group that consists of a carbonyl center (carbon -oxygen double bond) provided that the carbon atom is attached to hydrogen and R group of the carbon chain. A molecular formula depicts the chemical symbols for the elements constituting the given molecule such that each symbol contains a numerical subscript that describes the number of atoms of each atom present in the molecule.
Answer to Problem 15P

Explanation of Solution
To draw the structure of an aldehyde with the molecular formula

Want to see more full solutions like this?
Chapter 16 Solutions
GEN ORG + BIO (LL) W/CONNECT
- Dexamethasone is a halogen-containing steroid used to treat infl ammation in rheumatoid arthritis and other conditions. (a) Classify the alkyl halide in dexamethasone as 1 °, 2 °, or 3 °. (b) Classify the hydroxyl groups as 1 °, 2 °, or 3 °.arrow_forward1. What functional group is produced when an aldehyde reacts with H2/Pt? A.secondary alcohol B. carboxylic acid C.hemiacetal D. primary alcohol E.alkane F.tertiary alcohol G. alkene 2. What reaction occurs when an aldehyde reacts with H2/Pt to form a primary alcohol? A. Hydration B. Hydration C. Dehydration D. Oxidation E. Reduction( hydrogentation) 3. What reaction occurs when an Ester react with H+/H2O to from a carboxylic acid and alcohol? A. Dehydration B. Reduction ( Hydrogenation) C.Hydrolysis D. Hydration E.oxidationarrow_forwardDraw the structure of a compound fitting each description: a.an aldehyde with molecular formula C4H8O b. ketone with molecular formulab C4H8O c. a carboxylic acid with molecular formula C4H8O2 d. an ester with molecular formula C4H8O2arrow_forward
- 6. Draw the correct structures for the following: a. what is the correct structure of 3-methyl 1-pentyne? b. what is the correct structure of butyl methyl amine? c. what is the correct structure of 3-methyl 1-pentyne? d. what is the correct structure of 2-methyl 2-butanol? e. what is the correct structure of pentanoic acid?arrow_forward1) What is the preferred IUPAC name of the compound in Figure 12. [Numbers are separated by commas and numbers and letters by hyphens. Use lowercase letters and do not use spaces.] 2) Why is the structure in Figure 13 not numbered correctly? * a) Numbering should be started from the opposite end. b) The longest continuous chain was not selected. c) The sum rule was not used correctly when numbering this compound. d) The alphabet rule should be used when numbering this compound. e) The longest chain does not have the simplest side chains.arrow_forward(a) Draw the four isomers of C₅H₁₀O that can be oxidizedto an aldehyde. (b) Draw the three isomers of C₅H₁₀O that canbe oxidized to a ketone. (c) Draw the isomers of C₅H₁₀O that cannot be easily oxidized to an aldehyde or ketone. (d) Name any isomer that is an alcohol.arrow_forward
- an acetal is a combination of what functional group a)an ester and an ether b)an ether and a hydroxyl , bonded to adjacent carbons c)two ethers d) an ether and a hydroxyl, bonded to the same carbonarrow_forwardDraw the structure of the six constitutional isomers of molecular formula C 5H 12O that contain an ether as functional group.arrow_forward1. Which statement is correct? A. Aldehydes and ketones can hydrogen bond with other aldehydes and ketones. B. Aldehydes and ketones can hydrogen bond with water. C. Aldehydes and ketones cannot hydrogen bond with water. D. Aldehydes and ketones are always soluble in water.arrow_forward
- Identify the IUPAC name of the given structure. A. 2 - methylhexan-5-one B. 5 - methylhexan-2-one C. 2 - heptanone D. 5 - heptanone Identify the IUPAC name of the given structure. A. 4 - bromopentan-3-one B. 1 - bromobutan-2-one C. 2 - bromobutan-one D. None of the abovearrow_forwardA-AgNO3 B-BaCl2 C-CuSO4 D-HCl E-H2SO4 F-NaI G-NaBr H-NaOH I-(NH4)2SO4 J-NH4Cl Explain your answerarrow_forward1-Octen-3-ol is a potent mosquito attractant commonly used in mosquito traps. A number of reactions, including hydrogenation, will transform 1-octen-3-ol into a less effective molecule. Draw the structure of a hydrogenation product of 1-octen-3-ol.arrow_forward
 Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning




