
(a)
Interpretation:
Among the given designations, the one or more which may be correct for the given situation has to be chosen.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
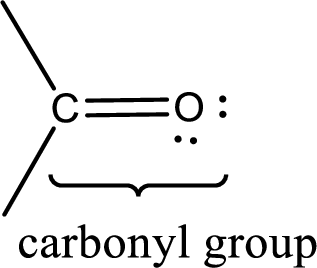
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
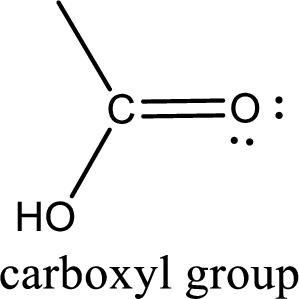
Carboxylic acid derivatives are the ones that are synthesized from or converted to a carboxylic acid. The generalized structural representation of carboxylic acid derivatives is shown below,
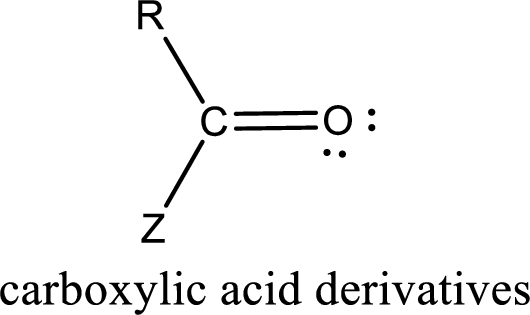
Carbon bonded to an oxygen atom through double bond is known as carbonyl group. The carbonyl group bonded to a R group means it is known as acyl group. If the acyl group is bonded to oxygen, nitrogen or halogen atom then the compound is known as acyl compound. If the acyl group is bonded to a carbon or hydrogen atom, then the compound is known as carbonyl compound. If the carbonyl carbon atom is bonded to a hydroxyl group then it is a carboxylic acid.
(b)
Interpretation:
Among the given designations, the one or more which may be correct for the given situation has to be chosen.
Concept Introduction:
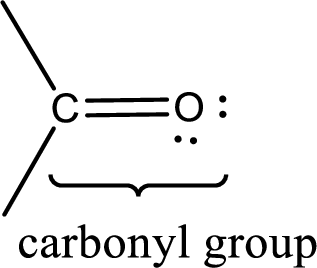
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
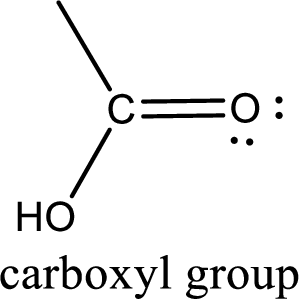
Carboxylic acid derivatives are the ones that are synthesized from or converted to a carboxylic acid. The generalized structural representation of carboxylic acid derivatives is shown below,
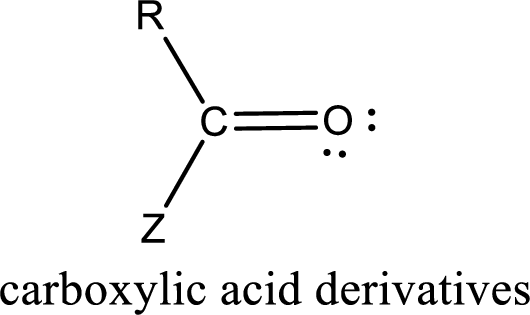
Carbon bonded to an oxygen atom through double bond is known as carbonyl group. The carbonyl group bonded to a R group means it is known as acyl group. If the acyl group is bonded to oxygen, nitrogen or halogen atom then the compound is known as acyl compound. If the acyl group is bonded to a carbon or hydrogen atom, then the compound is known as carbonyl compound. If the carbonyl carbon atom is bonded to a hydroxyl group then it is a carboxylic acid.
(c)
Interpretation:
Among the given designations, the one or more which may be correct for the given situation has to be chosen.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
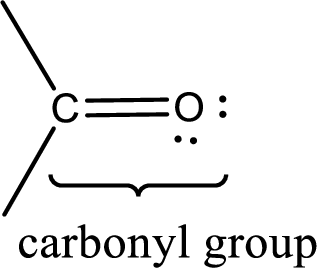
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
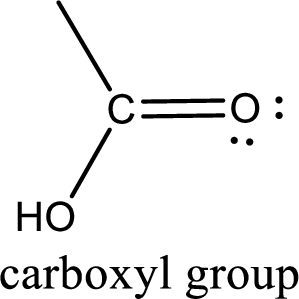
Carboxylic acid derivatives are the ones that are synthesized from or converted to a carboxylic acid. The generalized structural representation of carboxylic acid derivatives is shown below,
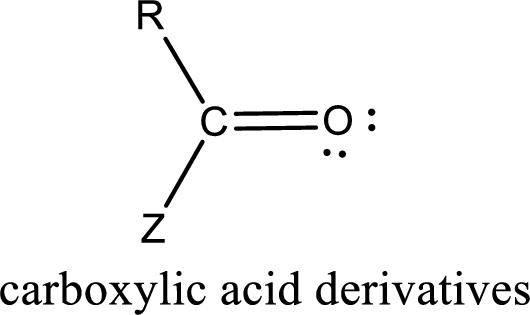
Carbon bonded to an oxygen atom through double bond is known as carbonyl group. The carbonyl group bonded to a R group means it is known as acyl group. If the acyl group is bonded to oxygen, nitrogen or halogen atom then the compound is known as acyl compound. If the acyl group is bonded to a carbon or hydrogen atom, then the compound is known as carbonyl compound. If the carbonyl carbon atom is bonded to a hydroxyl group then it is a carboxylic acid.
(d)
Interpretation:
Among the given designations, the one or more which may be correct for the given situation has to be chosen.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
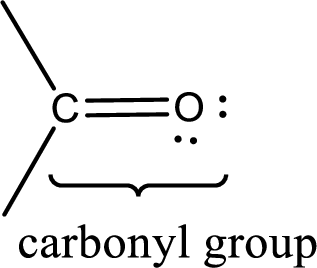
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
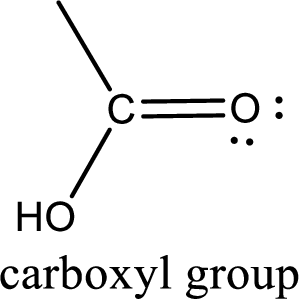
Carboxylic acid derivatives are the ones that are synthesized from or converted to a carboxylic acid. The generalized structural representation of carboxylic acid derivatives is shown below,
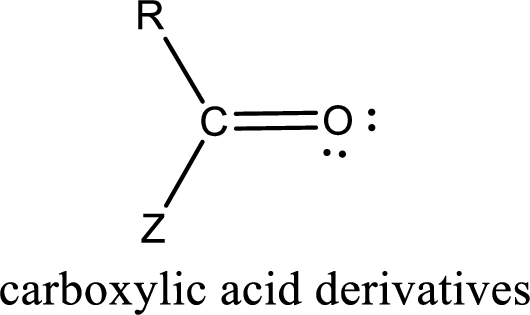
Carbon bonded to an oxygen atom through double bond is known as carbonyl group. The carbonyl group bonded to a R group means it is known as acyl group. If the acyl group is bonded to oxygen, nitrogen or halogen atom then the compound is known as acyl compound. If the acyl group is bonded to a carbon or hydrogen atom, then the compound is known as carbonyl compound. If the carbonyl carbon atom is bonded to a hydroxyl group then it is a carboxylic acid.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
General, Organic, and Biological Chemistry
- Need help determining the acidic and neutral components in an unknown sample. Have one of the following functional groups: Aldehyde Amine Ketone Alcohol Carboxylic acid Ester Phenol May have: Chlorine Ether Bromine Alkene lodine Alkyne Nitro Aromaticarrow_forwardDraw the all the reactants and products involved in a condensation reaction between propanoic acid and ethanamine. Make sure to include the catalyst needed and any secondary products that would form.arrow_forwardPlease name the following carboxylic acid or ester (spell out the full name of the compound) :) A) refer to first photo B) refer to second photoarrow_forward
- When some amount of strong acid is added to a solution of benzoic acid,________________arrow_forwardExplain why NOT ALL hydrolysis of esters produce a sour smellarrow_forwardEach of the following acids contains an additional type of functional group besides the carboxyl group. For each acid, specify the non-carboxyl functional group present. 9. Acrylic acid10. Lactic acid11. Maleic acid12. Glycolic acid13. Fumaric acid14. Pyruvic acid15. Malic acid16. Tartaric acidarrow_forward
- Acid Alcohol Odor Structure salicylic acid methanol wintergreen ? anthranilic acid methanol Grape ?arrow_forwardBriefly explain the following. Why hydrolysis of esters produce a sour smell in some reactions? Why are alcohol solutions used in hydroxamic test for esters?arrow_forwardTrue or false: An aldehyde is stronger than acid than carboxylic acid because it loses proteins thus resulting into a more stable ion.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,


