
Concept explainers
Interpretation:
The structure for the compound having molecular formula
Concept introduction:
In
In addition to chemical shift, a
Complicated splitting patterns can result when a proton is unequally coupled to two or more protons that are different from one another.
The ideal range for alkane protons is
Answer to Problem 16.31P
The proposed structure for the compound having molecular formula
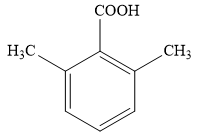
Explanation of Solution
The molecular formula for the given unknown compound is
The molecular formula shows an index of hydrogen deficiency of five.
The
IHD of five and the doublet at
The broad singlet at
The singlet at
For the aromatic protons, the signal at
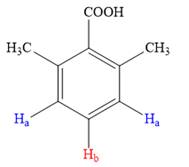
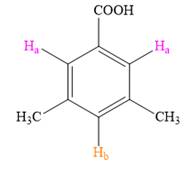
Both the trisubstituted benzenes have only two types of aromatic protons,
In the structure on left, the protons highlighted in blue will appear as a doublet as both of them have identical chemical environment, while the proton in red will split into a triplet as it has two neighboring protons attached.
In the structure on right, the protons in pink will appear as singlets, as they do not have any neighboring protons while the proton in orange will also appear as a singlet.
Thus, the second structure doesn’t match with the data given in the
The
The
The signal
Considering both the
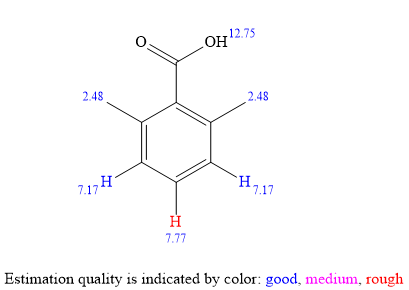
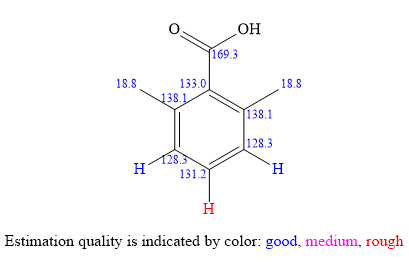
The structure of the unknown compound can be proposed based on its
Want to see more full solutions like this?
Chapter 16 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- 4) Draw the compound with molecular formula C9H100 that produces the following signals. Show your work by labeling your structure (Ha, Hb, etc.): 1H NMR a) 9.8 ppm, singlet, 1 H b) 7.3-7.6 ppm, multiplet (overlapping signals), 5 H c) 3.2 ppm, triplet, 2 H d) 2.8 ppm, triplet, 2H 13C NMR: 201.5, 140.7, 128.6, 128.5, 126.4, 45.2, 25.1 ppmarrow_forward1Compound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively. Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forwardCompound 1 has molecular formula C6H12. It shows three signals in the 1H-NMR spectrum, one at 0.96 ppm, one at 2.03 ppm, and one at 5.33 ppm. The relative integrals of these three signals are 3, 2, and 1, respectively. Compound 2 has molecular formula C7H15Br. It shows two signals in the 1H-NMR spectrum, one at 1.08 ppm and one at 1.59 ppm. The relative integrals of these two signals are 3 and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forward
- Which of the following compounds fits the proton NMR shown below? 2H 4.0 || A) CH₂CH₂CH₂COCH 3 || B) CH3CH₂COCH₂CH3 0 || C) CH3COCH₂CH₂CH3 0 || D) CH3CCH₂CH₂OCH₂ 3.0 3H 2.0 2H 3H 1.0 ppmarrow_forwardThe 1H-NMR spectrum of an unknown compound shows two signals, each of which is a singlet. Explaining your choice, predict which of compounds A-D could the unknown could be?arrow_forwardCompound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively. Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forward
- The 1H NMR spectrum of 2-propen-1-ol is shown here. Indicate the protons in the molecule that are responsible for each of the signals in the spectrum.arrow_forwardProblem: Predict the number of 'H NMR signals and the splitting pattern of each. (а) СНз СCH2 CНз (b) CH3 CH2 CCH2 CH3 (c) CH3 CCH(CH3)2arrow_forwardAre there signals that you can assign to specific protons in the molecule (labeled a-k)? Complete this column of the table. It may not be possible to assign each signal to a specific proton.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





