
Concept explainers
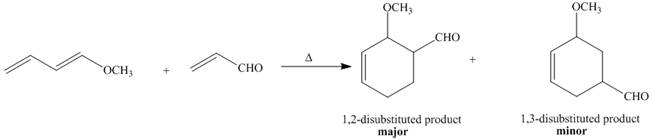
Diels–Alder reaction of a monosubstituted diene (such as
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
ORGANIC CHEMISTRY W/ ACCESS >IC<
- What product(s) would be obtained from the Diels-Alder reaction of transCH3CH=CHCO2Et with EACH of the following dienes:(a) 1.3- Butadiene(b) Isoprene(c) Cyclopentadiene________________________________arrow_forwardHow many linear dienes have molecular formula C6H10? (Disregard cis–trans isomers.) A. How many of the linear dienes in part a are conjugated dienes? B. How many are isolated dienes? C. How many are cumulated dienes?arrow_forwardThe pentadienyl radical, H2C“CH¬CH“CH¬CH2#, has its unpaired electron delocalized over three carbon atoms.(a) Use resonance forms to show which three carbon atoms bear the unpaired electron.(b) How many MOs are there in the molecular orbital picture of the pentadienyl radical?(c) How many nodes are there in the lowest-energy MO of the pentadienyl system? How many in the highest-energy MO?(d) Draw the MOs of the pentadienyl system in order of increasing energy. (continued)762 CHAPTER 15 Conjugated Systems, Orbital Symmetry, and Ultraviolet Spectroscopy(e) Show how many electrons are in each MO for the pentadienyl radical (ground state).(f) Show how your molecular orbital picture agrees with the resonance picture showing delocalization of the unpairedelectron onto three carbon atoms.(g) Remove the highest-energy electron from the pentadienyl radical to give the pentadienyl cation. Which carbon atomsshare the positive charge? Does this picture agree with the resonance picture?(h) Add an…arrow_forward
- Classify the following dienes and polyenes as isolated, conjugated, cumulated, or some combination of theseclassifications.(a) cycloocta-1,4-diene (b) cycloocta-1,3-dienearrow_forwardDefine Electrophilic addition and the Hammond postulate ?arrow_forwardClassify the following dienes and polyenes as isolated, conjugated, cumulated, or some combination of theseclassifications.(a) cycloocta-1,4-diene (b) cycloocta-1,3-diene (c) cyclodeca-1,2-diene(d) cycloocta-1,3,5,7-tetraene (e) cyclohexa-1,3,5-triene (benzene) (f) penta-1,2,4-trienearrow_forward
- The Diels–Alder reaction, a powerful reaction, occurs when a 1,3-diene such as A reacts with an alkene such as B to form the six-membered ring in C. Question: What bonds are broken and formed in this reaction?arrow_forwardplease compare and contrast the hybridization of ethylene and butadiene: ethylene butadiene LUMO LUMO HMO HMO Occupancy: Occupancy: Orbital energy (eV) Orbital energy (eV) Diels alder reactivity Diels alder reactivityarrow_forward7 The pentadienyl radical, H2C“CH¬CH“CH¬CH2 ) Draw the MOs of the pentadienyl system in order of increasing energyarrow_forward
- 1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. What is the theoretical yield of 2,4-pentanediol expressed in grams? Show calculations. What is the percentage yield of pure 2,4-pentanediol?arrow_forward1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. a. Provide a balanced chemical equation to show the reaction between 1,4-pentadiene and sulfuric acid. Do not use molecular formulas in the chemical equation except for sulfuric acid. b. What reactant is the limiting reagent in this chemical equation? Show calculations to support your answer.arrow_forwardDraw resonance forms to show how the BHA radical is stabilized by delocalization of the radical electron over other atoms in the molecule.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


