
Concept explainers
Interpretation:
It should be determined that which of the two
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
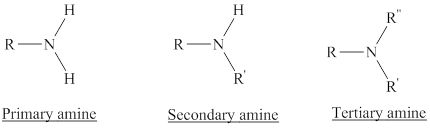
Hydrogen bond is an attractive force established between hydrogen atom attached to a highly electronegative element and another highly electronegative element of the same or different molecule.
High solubility is expected when hydrogen bonds form between solute and solvent molecule.
Solubility will decrease with increase in alkyl groups in a compound.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Fundamentals Of General, Organic, And Biological Chemistry Volume 1 Second Custom Edition For Washington State University, 2/e
- Propanamide and methyl acetate have about the same molar mass, both are quite soluble in water, and yet the boiling point of propanamide is 486 K, whereas that of methyl acetate is 330 K. Explain.arrow_forwardWhat product is formed when a solution of A and B is treated with mild base? This reaction is the first step in the synthesis of rosuvastatin (sold as a calcium salt under the trade name Crestor), a drug used to treat patients with high cholesterol.arrow_forwardCellulose is water insoluble, despite its many OH groups. Based on its three-dimensional structure, why do you think this is so?arrow_forward
- A pentapeptide has a titration curve that shows five inflection points for five equivalents of hydroxide added. How many residues have ionizable groups within their side chains (R-groups) based on this information?arrow_forwardWhy are some solutes soluble in water and solutes soluble in cyclohexane ?arrow_forwardWhich one of the following substances would you expect to be most soluble in water?A) ButanolB) Butanoic acidC) 2-pentanoneD) Propanearrow_forward
