
Concept explainers
(a)
Interpretation:
The products should be identified by the reaction of 2-heptanone with
Concept Introduction:
Addition of H2gas to a multiple bond is known as hydrogenation. In the presence of palladium metal as the catalyst, H2 molecules react with
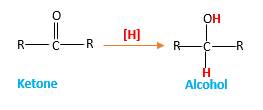
Answer to Problem 16.87P
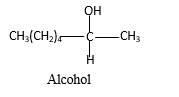
Explanation of Solution
When a ketone reacts with H2 gas in the presence of palladium metal resulting product is the secondary alcohol of the initial ketone molecule. Palladium metal act as a catalyst to the reaction that provides a surface to bind both the H2 and carbonyl compound which reduce the activation energy of the reaction.
Hydrogen atoms in the alcohol molecule shown below, which are indicated in red color are the added H during the hydrogenation reaction.
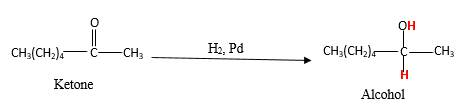
(b)
Interpretation:
The products should be identified by the reaction of 2-heptanone with
Concept Introduction:
Addition of an O atom in to a molecule is known as oxidation. If a carbonyl atom consists of a hydrogen atom directly connected to the carbonyl C, it will be oxidized in the presence of an oxidizing agent such as
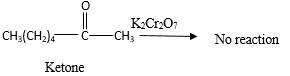
Answer to Problem 16.87P
No reaction.
Explanation of Solution
Addition of an O atom in to a molecule is known as oxidation. If a carbonyl atom consists of a hydrogen atom directly connected to the carbonyl C, it will be oxidized in the presence of an oxidizing agent such as
Hence, during the reaction no color change can be observed.
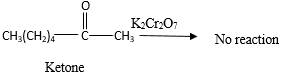
(c)
Interpretation:
The products should be identified by the reaction of 2-heptanone with
Concept Introduction:
Addition of an O atom in to a molecule is known as oxidation. If a carbonyl atom consists of a hydrogen atom directly connected to the carbonyl C, it will be oxidized in the presence of an oxidizing agent such as (
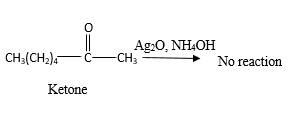
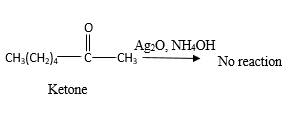
Answer to Problem 16.87P
No reaction.
Explanation of Solution
Addition of an O atom in to a molecule is known as oxidation. If a carbonyl atom consists of a hydrogen atom directly connected to the carbonyl C, it will be oxidized in the presence of an oxidizing agent such as
Hence, during the reaction is no silver mirror can be observed.
(d)
Interpretation:
The products should be identified by the reaction of 2-heptanone with
Concept Introduction:
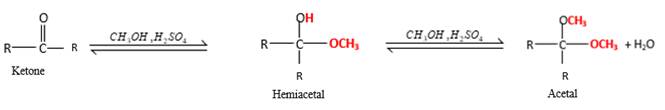
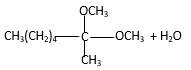
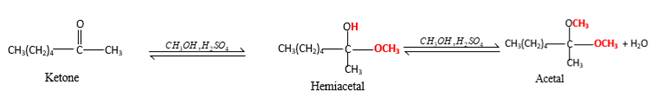
Inthe presence of alcohol in the acidic medium, ketones undergo addition reactions and give acetal in two steps.
Hydrogen atom and CH3 groups in the acetal and hemiacetal molecules shown below, which are indicated in red color are the added molecules during the reaction.
Answer to Problem 16.87P
Explanation of Solution
In the presence of alcohol in the acidic medium, ketones undergo addition reactions and give acetal in two steps. In the first step ketones form hemiacetals and during the second step it converts to an acetal molecule of the respective ketone molecule.
Addition of one molecule of alcohol in to a ketone forms a hemiacetal, one bond of the
Hydrogen atom, CH3 and OCH3 groups in the acetal and hemiacetal molecules shown below, which are indicated in red color are the added molecules during the reaction.
(e)
Interpretation:
The products should be identified by the reaction of 2-heptanone with
Concept Introduction:
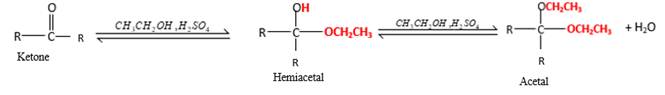
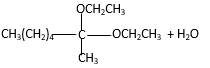
In the presence of alcohol in the acidic medium,
Hydrogen atom and CH2CH3 groups in the acetal and hemiacetal molecules shown below, which are indicated in red color are the added molecules during the reaction.
Answer to Problem 16.87P
Explanation of Solution
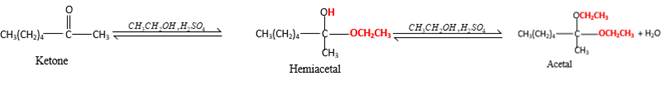
In the presence of alcohol in the acidic medium, ketones undergo addition reactions and give acetal in two steps. In the first step ketones form hemiacetals and during the second step it converts to an acetal molecule of the respective ketone molecule.
Addition of one molecule of alcohol in to aketone forms a hemiacetal, one bond of the
Hydrogen atom, CH2CH3 and OCH2CH3 groups in the acetal and hemiacetal molecules shown below, which are indicated in red color are the added molecules during the reaction.
(f)
Interpretation:
The products should be identified by the reaction of
Concept Introduction:
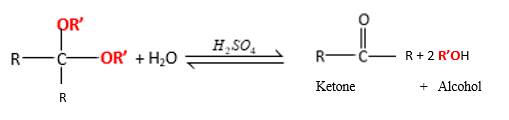
In the presence of waterandacid, acetals undergo hydrolysis reaction and produce aldehydes.
OR' groups in the acetal molecule shown below, which are indicated in red color are the molecules which becomes alcohol molecules during the hydrolysis.
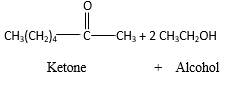
Answer to Problem 16.87P
Explanation of Solution
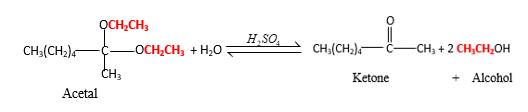
Acetals are stable molecules, but their bonds can cleave by a reaction with water and produce aldehydes.
In the acetal molecule, two bonds of the
CH2CH3 groups in the acetal molecule shown below, which are indicated in red color are the molecules which becomes alcohol molecules during the hydrolysis.
Want to see more full solutions like this?
Chapter 16 Solutions
GENERAL ORGANIC+BIOCHEM (LL)W/CONNECT
- Draw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b. CH3COCl c. (CH3CO)2O d. excess CH3I e. (CH3)2C = O f. CH3COCl, AlCl3 g. CH3CO2H h. NaNO2, HCl i. Part (b), then CH3COCl, AlCl j. CH3CHO, NaBH3CNarrow_forwardDraw the products formed when D-altrose is treated with each reagent. a. (CH3)2CHOH, HCl b. NaBH4, CH3OH c. Br2, H2O d. HNO3, H2O e. [1] NH2OH; [2] (CH3CO)2O, NaOCOCH3; [3] NaOCH3 f. [1] NaCN, HCl; [2] H2, Pd-BaSO4; [3] H3O+ g. CH3I, Ag2O h. C6H5CH2NH2, mild H+arrow_forwardDraw the products formed when ethylene oxide is treated with each reagent. a. HBr b. H2O(H2SO4) c. [1] CH3CH2O; [2] H2O d. [1] HC ≡ C−; [2] H2O e. [1] −OH; [2] H2O f. [1] CH3S−; [2] H2Oarrow_forward
- Which reagents are used for reaction 1? a NaN3, ethanol and NaBH4, ethanol, H3O+ b CH3CH2NH2, NaOH c NH3, NaOH d NH3, DCC Which reagent are used for reaction 2? a CH3COOH, DCC b CH3COCH3 and NaBH4, ethanol, H3O+ c CH3COH and NaBH4, ethanol, H3O+ d CH3CONH2 and CH3CH2NH2arrow_forwardOChem help with IUPAC names involving Ph and Bn The phenyl group (Ph-R, C6H5-R) can be formed by removing a hydrogen from benzene and attaching a substituent to where the hydrogen was removed. The benzyl group (abbv. Bn), similar to the phenyl group, is formed by manipulating the benzene ring. In the case of the benzyl group, it is formed by taking the phenyl group and adding a CH2 group to where the hydrogen was removed. Its molecular fragment can be written as C6H5CH2-R, PhCH2-R, or Bn-R. Please provide the IUPAC name for the following: (Ph)2CHC(CH3)2CC(CH2)3CH(Bn)CHOarrow_forwardDraw the missing starting material. Reagent 1 is benzene and AlCl3. Reagent B is Zn(Hg) and HCl.arrow_forward
- A. OsO4 and NMO B. Br2 and H20 C. Hg(OAc)2, H2O and NaBH4, NaOH D. RCO3H E. BH3-THF and H2O2, NaOH Which reagent will complete this reaction?arrow_forwardDraw the products formed when phenol (C6H5OH) is treated with each set of reagents. a. [1] HNO3, H2SO4; [2] Sn, HCl b. [1] (CH3CH2)2CHCOCl, AlCl3; [2] Zn(Hg), HCl c. [1] CH3CH2Cl, AlCl3; [2] Br2, hν d. [1] (CH3)2CHCl, AlCl3; [2] KMnO4arrow_forwardWhat is the reagent in this reaction? a. mCPBA, water, H+ b. H2, Pd c. NaBH4, water d. KMNO4arrow_forward
- (a) How will you convert:(i) Benzene to acetophenone (ii) Propanone to 2-Methylpropan-2-ol(b) Give reasons :(i) Electrophilic substitution in benzoic acid takes place at meta position.(ii) Carboxylic acids are higher boiling liquids than aldehydes, ketones and alcohols of comparable molecular masses.(iii) Propanal is more reactive than propanone in nucleophilic addition reactions.arrow_forwardWhat is the product of the reaction of 2-methyl-2-butene with Br2/H2O? 3-bromo-2-methyl-2-butanol 2-bromo-3-methyl-2-butanol 4-bromo-2-methyl-2-butanol 2,3-dibromo-2-methylbutanearrow_forwardGive reasons: (i) Bond length of C = O in carboxylic acids is slightly larger than C = O bond length in carbonyl compounds. (ii) There are two –NH2 groups in semicarbazide. However, only one –NH2 group is involved in the formation of semicarbazones. (iii) Benzoic acid is less soluble in water than acetic acid. (iv) Formic acid is a stronger acid than acetic acid.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
