
Concept explainers
dentify each of the following
NMR spectra:
Interpretation:
The structures of each of the
Concept introduction:
In
The number of signals in a spectrum gives information about the type of carbon atoms present in the structure of the compound.
In
The
Index of hydrogen deficiency (IHD) is calculated by the equation as follows:
Here,
Oxygen atoms do not disturb the index of hydrogen deficiency.
At
At
A methine is
Answer to Problem 38P
Solution: The structures of each of the
a)
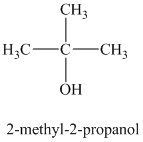
b)
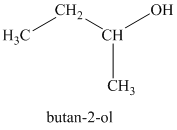
c)
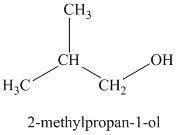
Explanation of Solution
a)
The molecular formula shows an index of hydrogen deficiency equal to zero. Thus, the alcohol contains no ring or multiple bonds. The
The signal
The signal
The structure must contain three equivalent methyl groups and one tertiary carbon atom. Therefore, the structure of this isomer is shown as follows:
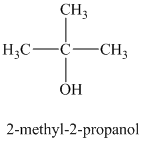
This isomer is a tertiary alcohol.
b)
The molecular formula shows an index of hydrogen deficiency equal to zero. Thus, the alcohol contains no ring or multiple bonds. The
The signal
The signal
The signal
Thus, the structure must contain two non-equivalent methyl groups, one methylene group, and one methine group to which the hydroxyl group is attached.
Therefore, the structure of this isomer is shown as follows:

This isomer is a secondary alcohol.
c)
The molecular formula shows an index of hydrogen deficiency equal to zero. Thus, the alcohol contains no ring or multiple bonds. The
The signal
The signal
The signal
Thus, the structure must contain two equivalent methyl groups, one methylene group, and one methine group to which the hydroxyl group is attached.
Therefore, the structure of this isomer is shown as follows:

Want to see more full solutions like this?
Chapter 16 Solutions
ORGANIC CHEMISTRY (LL)-W/SOLN.>CUSTOM<
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





