
CHEMISTRY:CENTRAL..(LL)W/S.G.+MASTERING
14th Edition
ISBN: 9780134773223
Author: Brown
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 16, Problem 43E
(a)
Interpretation Introduction
Interpretation: The 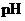 of
of 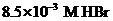 solution should be calculated.
solution should be calculated.
Concept Introduction:
The 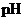 is calculated by the formula,
is calculated by the formula,
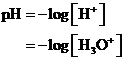
Where,
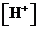 or
or 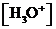 is the concentration of
is the concentration of 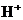 ions.
ions.
(b)
Interpretation Introduction
Interpretation: The 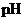 of
of 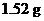
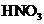 in
in 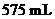 of solution should be calculated.
of solution should be calculated.
Concept Introduction:
The 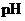 is calculated by the formula,
is calculated by the formula,
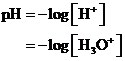
Where,
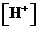 or
or 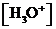 is the concentration of
is the concentration of 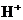 ions.
ions.
(c)
Interpretation Introduction
Interpretation: The 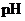 of
of 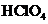 solution when the solution is diluted to
solution when the solution is diluted to 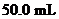 should be calculated.
should be calculated.
Concept Introduction:
The 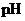 is calculated by the formula,
is calculated by the formula,
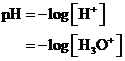
Where,
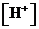 or
or 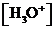 is the concentration of
is the concentration of 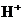 ions.
ions.
(d)
Interpretation Introduction
Interpretation: The 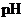 of a solution that is formed by the mixing
of a solution that is formed by the mixing 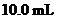 of
of 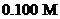
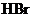 with
with 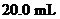 of
of 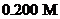
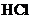 should be calculated.
should be calculated.
Concept Introduction:
The 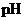 is calculated by the formula,
is calculated by the formula,
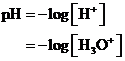
Where,
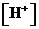 or
or 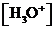 is the concentration of
is the concentration of 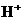 ions.
ions.
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 16 Solutions
CHEMISTRY:CENTRAL..(LL)W/S.G.+MASTERING
Ch. 16.2 - Practice Exercise 1 Consider the following...Ch. 16.2 - Prob. 16.1.2PECh. 16.2 - Prob. 16.2.1PECh. 16.2 - Practice Exercise 2 When lithium oxide (Li2O) is...Ch. 16.2 - Based on information in Figure 16.4, place the...Ch. 16.2 - Practice Exercise 2 For each reaction, use Figure...Ch. 16.3 - Practice Exercise 1 In a certain acidic solution...Ch. 16.3 - Practice Exercise 2 Indicate whether solutions...Ch. 16.3 - Prob. 16.5.1PECh. 16.3 - Prob. 16.5.2PE
Ch. 16.4 - Practice Exercise 1 A solution at 250C has [OH-] =...Ch. 16.4 - Practice Exercise 2 In a sample of lemon juice,...Ch. 16.4 - Practice Exercise 1 A solution at 25° C has pOH =...Ch. 16.4 - Prob. 16.7.2PECh. 16.5 - Practice Exercise 1 Order the following three...Ch. 16.5 - Prob. 16.8.2PECh. 16.5 - Practice Exercise 1 Order the following three...Ch. 16.5 - Prob. 16.9.2PECh. 16.6 - Prob. 16.10.1PECh. 16.6 - Practice Exercise 2 Niacin, one of the B vitamins,...Ch. 16.6 - Prob. 16.11.1PECh. 16.6 - Practice Exercise 2 A 0.020 M solution of niacin...Ch. 16.6 - Practice Exercise 1 What is the pH of a 0.40 M...Ch. 16.6 - Practice Exercise 2 The Ka for niacin (Sample...Ch. 16.6 - Prob. 16.13.1PECh. 16.6 - Prob. 16.13.2PECh. 16.6 - Practice Exercise 1 What is the pH of a 0.28 M...Ch. 16.6 - Practice Exercise 2 Calculate the pH of a 0.020 M...Ch. 16.7 - Prob. 16.15.1PECh. 16.7 - Practice Exercise 2 Which of the following...Ch. 16.7 - Prob. 16.16.1PECh. 16.7 - Practice Exercise 2 What is the morality of an...Ch. 16.8 - Practice Exercise 1 By using information from...Ch. 16.8 - Practice Exercise 2 Based on information in...Ch. 16.9 - Prob. 16.18.1PECh. 16.9 - Prob. 16.18.2PECh. 16.9 - Practice Exercise 1 How many of the following...Ch. 16.9 - Practice Exercise 2 Predict whether the...Ch. 16.10 - Prob. 16.20.1PECh. 16.10 - In each pair, choose the compound that gives the...Ch. 16 - Prob. 1DECh. 16 - a. Identify the Br ted-Lowry acid and base in the...Ch. 16 - Prob. 2ECh. 16 - Prob. 3ECh. 16 - Prob. 4ECh. 16 - 16.5 The following diagrams represent aqueous...Ch. 16 - Prob. 6ECh. 16 - Which of these statements about how the percent...Ch. 16 - 16.8 Each of the three molecules shown here...Ch. 16 - Prob. 9ECh. 16 - Which of the following diagrams best represent an...Ch. 16 - Prob. 11ECh. 16 - Prob. 12ECh. 16 - Prob. 13ECh. 16 - 16.14 Which of the following statements is...Ch. 16 - Prob. 15ECh. 16 - Prob. 16ECh. 16 - Identify the Bronsted-Lowry acid and the...Ch. 16 - Prob. 18ECh. 16 - Prob. 19ECh. 16 - Prob. 20ECh. 16 - Prob. 21ECh. 16 - Prob. 22ECh. 16 - Prob. 23ECh. 16 - Prob. 24ECh. 16 - Prob. 25ECh. 16 - Prob. 26ECh. 16 - Prob. 27ECh. 16 - Prob. 28ECh. 16 - 16.29 Calcualte [H +] for each of the following...Ch. 16 - Prob. 30ECh. 16 - 16.31 At the freezing point of water (0 o C), K10...Ch. 16 - Prob. 32ECh. 16 - Prob. 33ECh. 16 - Prob. 34ECh. 16 - 16.35 Complete the following table by calculating...Ch. 16 - Prob. 36ECh. 16 - Prob. 37ECh. 16 - 16.38 Carbon dioxide in the atmosphere dissolves...Ch. 16 - Prob. 39ECh. 16 - Prob. 40ECh. 16 - Prob. 41ECh. 16 - Prob. 42ECh. 16 - Prob. 43ECh. 16 - Prob. 44ECh. 16 - Prob. 45ECh. 16 - Prob. 46ECh. 16 - Prob. 47ECh. 16 - Prob. 48ECh. 16 - Prob. 49ECh. 16 - write the chemical equation and the Ka expression...Ch. 16 - Prob. 51ECh. 16 - Prob. 52ECh. 16 - Prob. 53ECh. 16 - Prob. 54ECh. 16 - Prob. 55ECh. 16 - Prob. 56ECh. 16 - Prob. 57ECh. 16 - Prob. 58ECh. 16 - Calculate the pH of each of the following solution...Ch. 16 - Prob. 60ECh. 16 - Prob. 61ECh. 16 - Prob. 62ECh. 16 - Calculate the percent ionization of hydrazoic acid...Ch. 16 - 16.64 Calculate the percent ionization of...Ch. 16 - Prob. 65ECh. 16 - Prob. 66ECh. 16 - Prob. 67ECh. 16 - 16.68 The hypochlorite ion, CIO- , acts as a weak...Ch. 16 - Prob. 69ECh. 16 - Prob. 70ECh. 16 - Calculate the molar concentration of OH- in a...Ch. 16 - 16.72 Calculate the molar concentration of OH- in...Ch. 16 - Prob. 73ECh. 16 - Prob. 74ECh. 16 - Prob. 75ECh. 16 - Prob. 76ECh. 16 - a. Given that Ka for acetic acid is 1.8 10-5 and...Ch. 16 - 16.78
a. Given that Kb for ammonia is 1.8 X 10 -5...Ch. 16 - Prob. 79ECh. 16 - Prob. 80ECh. 16 - Prob. 81ECh. 16 - Pyridinium bromide (C5H5NHBr) is a strong...Ch. 16 - Prob. 83ECh. 16 - Prob. 84ECh. 16 - Prob. 85ECh. 16 - 16.86 An unknown salt is either KBr, NH4 C1, KCN,...Ch. 16 - Prob. 87ECh. 16 - Prob. 88ECh. 16 - 16.89 Based on their compositions and structures...Ch. 16 - Prob. 90ECh. 16 - 16.91 Indicate whether each of the following...Ch. 16 - Prob. 92ECh. 16 - Prob. 93ECh. 16 - Prob. 94ECh. 16 - Prob. 95ECh. 16 - Prob. 96ECh. 16 - Prob. 97ECh. 16 - Prob. 98ECh. 16 - Prob. 99AECh. 16 - Prob. 100AECh. 16 - Prob. 101AECh. 16 - Prob. 102AECh. 16 - Prob. 103AECh. 16 - Prob. 104AECh. 16 - Benzoic acid (C6H5COOH) and aniline (C6H5NH2) are...Ch. 16 - Prob. 106AECh. 16 - Prob. 107AECh. 16 - Prob. 108AECh. 16 - Butyric acid is responsible for the foul smell of...Ch. 16 - Prob. 110AECh. 16 - Prob. 111AECh. 16 - Prob. 112AECh. 16 - 1S.113 Many moderately large organic molecules...Ch. 16 - Prob. 114AECh. 16 - Prob. 115AECh. 16 - Prob. 116IECh. 16 - Prob. 117IECh. 16 - Prob. 118IECh. 16 - Prob. 119IECh. 16 - 16.120 At 50 oC, the ion-product constant for H2...Ch. 16 - Prob. 121IECh. 16 - Prob. 122IECh. 16 - Prob. 123IECh. 16 - Prob. 124IECh. 16 - Prob. 125IECh. 16 - Prob. 126IE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY