
Concept explainers
(a)
Interpretation:
The structure of the salmeterol should be drawn.
Concept Introduction:
(b)
Interpretation:
One chirality center in a salmeterol molecule should be identified.
Concept Introduction:
Chirality is the presence of an asymmetric carbon center in a molecule and a molecule which contains a chiral center cannot superimpose on its mirror image. To consider as chiral, molecule or object and its mirror image should not superimpose. To consider as achiral, molecule or object and its mirror image should be superimposed with each other.
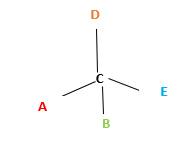
In the above diagram, where C is the chiral center/ asymmetric carbon center.
A, B, D, E are four different functional groups.
(c)
Interpretation:
Enantiomers of salmeterol in three dimensions should be drawn.
Concept Introduction:
Mirror image of a molecule is a reflected duplicate of the molecule.
Enantiomers − the mirror image of the original molecule of a chiral molecule. These are stereoisomers.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
- Draw the structure for a compound with molecular formula C2H2I2F2 a. that is optically inactive because it does not have an asymmetric center. b. that is optically inactive because it is a meso compound. c. that is optically active.arrow_forwardLocate the stereogenic center in each compound and draw both enantiomers. b. HO, OH С. HO. NH2 a.arrow_forwardClassify each compound as IDENTICAL to A or an ENANTIOMER to A.arrow_forward
- Ephedrine, a stimulant and illegal drug is an optically active compound with two chiral centers. OH NHCH3 CH3 Ephedrine a. In the structure of ephedrine on the answer sheet, mark each chiral center with an asterisk. b. How many stereoisomer/s does ephedrine have? Show necessary solutions on the answer sheet. c. Compound EPS is a stereoisomer of ephedrine. Determine the absolute configuration (R or S) at carbons A and B of compound EPS.arrow_forwardDraw the mirror image of each compound. Label each molecule as chiral or achiral.arrow_forward4. ) Determine if the following compound is chiral or not. OH HO OH Me ZOH Ме HO HO OH F. CI Ме CI Он Me JCHO CI Br Br CI CH3arrow_forward
- Locate the stereogenic centers in each molecule. Compounds may have one or more stereogenic centers.arrow_forward2. What is the relationship between these two molecules? o a. Identical b. Constitutional isomers c. Enantiomers d. Diastereomersarrow_forwardLabel each compound as chiral or achiral.arrow_forward
- 4. Two stereoisomers (G) and (H) are obtained from the reaction of HBr with alkene F. Draw the structures of G and H, showing the stereochemistry at the new chiral centre. What is the relationship between these two stereoisomers? F Br HBr G + Harrow_forward#23arrow_forwardClassify each compound as identical to A or its enantiomer. CHO CHO H OH CHO a. CH,CH-C--OH CumCH,CH3 H OH b. d. HO C. ČH,CH3 н он CH;CH CHO CHO Aarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

