
OWL V2 with MindTap Reader and Student Solutions Manual eBook for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
8th Edition
ISBN: 9781305865617
Author: Brown, Iverson, Anslyn, FOOTE
Publisher: Cengage Learning US
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16.8, Problem BQ
The given mechanism of transamination reaction is shown below,
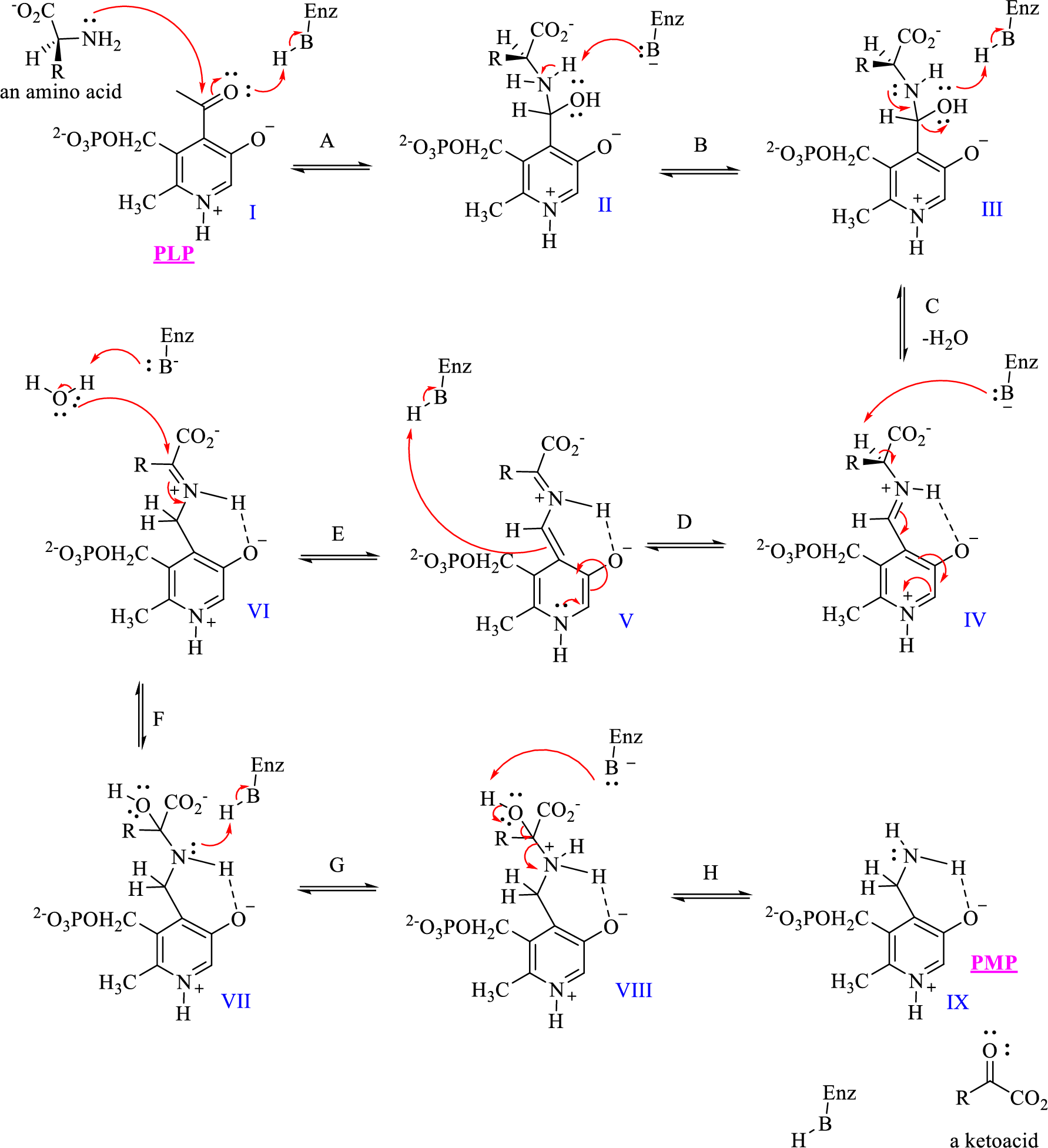
Which structures would be classified as tetrahedral intermediates derived from addition reactions?
- 1. II, III, VII, and VIII
- 2. IV, V, and VI
- 3. II, IV, and VIII
- 4. II and VI
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
37.The equilibrium constant that drives complexation reactions to completion is known as?
36.Eosin Y is also known as
What is the intermediate A for the following Stork enamine synthesis?
O
1.
H₂C.
CH3
A
2. H3O*
IZ
p-TSOH
(-H₂O)
O
CI
C12H1402
Identify the intermediate that is INITIALLY formed in an ozonolysis reaction.
03
A
B
||
C
IV
H H
D
IV
Chapter 16 Solutions
OWL V2 with MindTap Reader and Student Solutions Manual eBook for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
Ch. 16.1 - Write the IUPAC name for each compound. Specify...Ch. 16.1 - Write structural formulas for all aldehydes with...Ch. 16.1 - Write the IUPAC name for each compound.Ch. 16.5 - Prob. 16.4PCh. 16.6 - Prob. 16.5PCh. 16.7 - Prob. 16.6PCh. 16.7 - Write a mechanism for the acid-catalyzed...Ch. 16.8 - Prob. 16.8PCh. 16.8 - The given mechanism of transamination reaction is...Ch. 16.8 - The given mechanism of transamination reaction is...
Ch. 16.8 - The given mechanism of transamination reaction is...Ch. 16.8 - Prob. DQCh. 16.8 - Prob. EQCh. 16.8 - The given mechanism of transamination reaction is...Ch. 16.9 - Predict the position of the following equilibrium.Ch. 16.9 - Draw a structural formula for the keto form of...Ch. 16.10 - Prob. 16.11PCh. 16.11 - What aldehyde or ketone gives these alcohols upon...Ch. 16.11 - Prob. 16.13PCh. 16 - Prob. 16.14PCh. 16 - Prob. 16.15PCh. 16 - The infrared spectrum of compound A, C6H12O, shows...Ch. 16 - Following are 1H-NMR spectra for compounds B...Ch. 16 - Draw structural formulas for the product formed by...Ch. 16 - Suggest a synthesis for the following alcohols...Ch. 16 - Show how to synthesize the following alcohol using...Ch. 16 - 1-Phenyl-2-butanol is used in perfumery. Show how...Ch. 16 - Prob. 16.22PCh. 16 - Draw structural formulas for (1) the...Ch. 16 - Show how to bring about the following conversions...Ch. 16 - Prob. 16.25PCh. 16 - Wittig reactions with the following -chloroethers...Ch. 16 - Prob. 16.27PCh. 16 - Prob. 16.28PCh. 16 - 5-Hydroxyhexanal forms a six-membered cyclic...Ch. 16 - Prob. 16.30PCh. 16 - Prob. 16.31PCh. 16 - Propose a mechanism to account for the formation...Ch. 16 - Prob. 16.33PCh. 16 - Prob. 16.34PCh. 16 - Show how to bring about the following conversion.Ch. 16 - A primary or secondary alcohol can be protected by...Ch. 16 - Prob. 16.37PCh. 16 - Prob. 16.38PCh. 16 - Prob. 16.39PCh. 16 - Prob. 16.40PCh. 16 - The following molecule belongs to a class of...Ch. 16 - When cis-2-decalone is dissolved in ether...Ch. 16 - Prob. 16.43PCh. 16 - Prob. 16.44PCh. 16 - The following bicyclic ketone has two -carbons and...Ch. 16 - Propose a mechanism for this reaction.Ch. 16 - The base-promoted rearrangement of an -haloketone...Ch. 16 - If the Favorskii rearrangement of...Ch. 16 - (R)-Pulegone, readily available from pennyroyal...Ch. 16 - (R)-Pulegone is converted to (R)-citronellic acid...Ch. 16 - Starting with cyclohexanone, show how to prepare...Ch. 16 - Show how to convert cyclopentanone to these...Ch. 16 - Prob. 16.53PCh. 16 - Prob. 16.54PCh. 16 - Prob. 16.55PCh. 16 - Following is the structural formula of Surfynol, a...Ch. 16 - Prob. 16.57PCh. 16 - Propose a mechanism for this isomerization.Ch. 16 - Starting with acetylene and 1-bromobutane as the...Ch. 16 - Prob. 16.60PCh. 16 - Prob. 16.61PCh. 16 - Prob. 16.62PCh. 16 - Prob. 16.63PCh. 16 - Prob. 16.64PCh. 16 - All rearrangements we have discussed so far have...Ch. 16 - In dilute aqueous base, (R)-glyceraldehyde is...Ch. 16 - Treatment of -D-glucose with methanol in the...Ch. 16 - Treating a Grignard reagent with carbon dioxide...Ch. 16 - Prob. 16.69PCh. 16 - Prob. 16.70PCh. 16 - Prob. 16.71PCh. 16 - Prob. 16.72PCh. 16 - Write the products of the following sequences of...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Prob. 16.78PCh. 16 - Prob. 16.79PCh. 16 - Prob. 16.80PCh. 16 - Prob. 16.81P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine the de Brogue wavelength of a. an electron moving at 1/10 the speed of light. b. a 400 g Frisbee movi...
Inorganic Chemistry
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Give one example from main group chemistry that illustrates each of the following descriptions: (a) Covalent ne...
General Chemistry: Atoms First
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (14th Edition)
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (6th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Identify the intermediates (A-D) in the following reaction sequence.arrow_forward1. Enzyme + acetate + CoA → enzyme + acetyl-CoA In the reaction above, the enzyme is acting as a modulator cofactor catalyst activatorarrow_forwardWhich of A-E are intermediates that form along the pathway of the reaction shown below? Cl2, H2O/DMSO CI CI CI OH2 H2O CI CI O B and E O A and D O B and D O A and C O B and C MacBook Pro A.arrow_forward
- please answer all 34-36arrow_forwardSome microorganisms have evolved cellular machinery that carries out chemical transformations in a manner analogous to that of the pyruvate dehydrogenase complex (PDC). One example of these remarkable transformations is shown below. معلم H3C 1. TPP 2. lipoamide CH3 + 3. Coenzyme A H3C H H3C SCOA он Provide a complete curved-arrow mechanism for all three steps of this multistep enzymatic transformation. You may abbreviate the structure of Coenzyme A as "COA-SH" in your mechanism. You do not have to show a mechanism for the regeneration of the lipoamide cofactor.arrow_forward1. The enzyme that catalyzes the reversible reaction below requires what cofactor? A. Pyridoxal phosphate B. Paradoxal phosphate C. Tetrahydrofolate D. S-Adenosyl methionine 2. Skeletal muscle exports excess nitrogen to the liver in the form of... A. Alanine B. Tyrosine C. Pyruvate D. Glycine 3. In the urea cycle, ornithine transcarbamoylase condenses a ornithine with what molecule to produce citrulline? A. Carbamate B. Aspartate C. Phosphate D. Carbamoyl phosphatearrow_forward
- 9. The first two steps of ethanol metabolism are shown below. Which molecule is reduced over the course of these reactions? NAD* NADH+H* NAD* NADH+H* CH3CH2OH CHзCно CHзCOон alcohol ethanol dehydrogenase acetaldehyde acetic acid H20 acetaldehyde dehydrogenase a. ethanol and acetaldehyde b. alcohol dehydrogenase and acetaldehyde dehydrogenase c. ethanol and alcohol dehydrogenase d. acetic acid e. NAD+arrow_forwardWhich of the following reaction is mot likely to activate a drug molecule from orally administered inactive parent molecule? a Aromatic hydroxylation b Benzylic carbon oxidation c Hydrolysis d Azo reductionarrow_forwardThe dynamic process by which both forms of a- and B-D-glucopyranose in solution change slowly into an equilibrium mixture of both is known as (choose the name). OH OH Но но НО HO OH OH OH OH a-D-glucopyranose 36% B-D-glucopyranose 64% OA Acetylation O B. Enediol Rearrangement OC Mutarotation O D. Acetal Formation O E Epimerizationarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

DIGESTER-35 | VITAMINS AND THEIR RELATED COENZYMES| GPAT | NIPER | PHARMACIST| DI; Author: GPAT DISCUSSION CENTER;https://www.youtube.com/watch?v=CGrdNYmho0s;License: Standard YouTube License, CC-BY