
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 16.8A, Problem 16.10P
- a. Draw the molecular orbitals for the cyclopropenyl case.
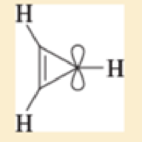
(Because there are three p orbitals, there must be three MOs one all-bonding MO and one degenerate pair of MOs.)
- b. Draw an energy diagram for the cyclopropenyl MOs. (The polygon rule is helpful.) Label each MO as bonding, nonbonding, or antibonding, and add the nonbonding line. Notice that it goes through the approximate average of the MOs.
- c. Add electrons to your energy diagram to show the configuration of the cyclopropenyl cation and the cyclopropenyl anion. Which is
aromatic and which is antiaromatic?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw one all-bonding MO, thena pair of degenerate MOs, and then a final pair of degenerate MOs. Draw the energydiagram, fill in the electrons, and confirm the electronic configurations of the cyclopentadienyl cation and anion
a-Draw a molecular orbital (MO) energy level diagram for NO. Label the atomic orbitals, MOs and identify the frontier orbitals.
b-Explain the differences in bond dissociation energies and bond lengths on addition and removalof an electron from NO.
Draw an energy diagram for the cyclopropenyl MOs. (The polygon rule is helpful.)Label each MO as bonding, nonbonding, or antibonding, and add the nonbonding line.Notice that it goes through the approximate average of the MOs.
Chapter 16 Solutions
Organic Chemistry (9th Edition)
Ch. 16.2 - Prob. 16.1PCh. 16.2 - Prob. 16.2PCh. 16.2 - a. Draw the resonance forms of benzene,...Ch. 16.2 - Show the product of the Diels-Alder dimerization...Ch. 16.4 - Prob. 16.5PCh. 16.6 - Make a model of cyclooctatetraene in the tub...Ch. 16.6 - Prob. 16.7PCh. 16.6 - Prob. 16.8PCh. 16.7 - Prob. 16.9PCh. 16.8A - a. Draw the molecular orbitals for the...
Ch. 16.8A - Repeat Problem16-10 for the cyclopentadienyl ions....Ch. 16.8C - Explain why each compound or ion should be...Ch. 16.8C - The following hydrocarbon has an unusually large...Ch. 16.8C - Prob. 16.14PCh. 16.8C - Prob. 16.15PCh. 16.9B - Prob. 16.16PCh. 16.9C - Show which of the nitrogen atoms in purine are...Ch. 16.9C - The proton NMR spectrum of 2-pyridone gives the...Ch. 16.9D - Prob. 16.19PCh. 16.9D - Prob. 16.20PCh. 16.10 - Prob. 16.21PCh. 16.12 - Ciprofloxacin is a member of the fluoroquinolone...Ch. 16.13 - Draw and name all the chlorinated benzenes having...Ch. 16.13 - Name the following compounds:Ch. 16.15 - The UV spectrum of 1-phenylprop-2-en-1-ol shows an...Ch. 16 - Prob. 16.26SPCh. 16 - Name the following compounds:Ch. 16 - Draw and name all the methyl, dimethyl, and...Ch. 16 - Four pairs of compounds are shown. In each pair,...Ch. 16 - One of the following hydrocarbons is much more...Ch. 16 - In Kekuls time cyclohexane was unknown, and there...Ch. 16 - Prob. 16.32SPCh. 16 - Azulene is a deep-blue hydrocarbon with resonance...Ch. 16 - Prob. 16.34SPCh. 16 - Prob. 16.35SPCh. 16 - Prob. 16.36SPCh. 16 - Prob. 16.37SPCh. 16 - Prob. 16.38SPCh. 16 - Prob. 16.39SPCh. 16 - Biphenyl has the following structure. a. Is...Ch. 16 - Anions of hydrocarbons are rare, and dianions of...Ch. 16 - How would you convert the following compounds to...Ch. 16 - Prob. 16.43SPCh. 16 - Prob. 16.44SPCh. 16 - A student found an old bottle labeled thymol on...Ch. 16 - Prob. 16.46SPCh. 16 - Prob. 16.47SPCh. 16 - Prob. 16.48SPCh. 16 - The proton NMR chemical shifts of the hydrogens in...Ch. 16 - Prob. 16.50SPCh. 16 - NMR has been used to probe many molecular...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the MO diagram of linear NO2 by consulting the character tables and orbital energies, and to align your O/N/O axes. Remember to use a lower symmetry point group for the linear molecule. What is the bond order of linear NO2? How does it change if you make it NO2 - or NO2 +. Draw each SALC and look for interactions between the orbitals.arrow_forwardDraw the d Orbital Energy Diagrams for Weak (1st pic) and Strong Field (2nd pic) Cases. Include the CFSE and uS for each.arrow_forwardWhich MOs are the HOMO and the LUMO in the excited state?arrow_forward
- 3) Combine any Atomic orbitals to form Molecular orbitals assuming they fulfill the phase and energy requirement. Construct MO diagram (include AOs, MOs, pictures of each, electrons, HOMO, LUMO, B.O., and comparative energies, etc.). REMOVE an ELECTRON to form CATION Please only do part 3. The first image shows the problemarrow_forwardDraw the complete MO diagram for [CoCl6]4- Label the AOs and SALCs and MOs. Draw a sketch for each MO with the appropriate symmetry labelarrow_forwardLet us construct the molecular orbital diagram of ethylene (in pieces). a. First, construct the MO diagram of linear carbene (CH2). Draw pictures of all 6 orbitals b. Now bend the carbene to a bond angle of about 120°. How does this change your MO diagram? Draw pictures of all 6 orbitals. c. Now bring two of these carbene molecules together to make ethylene. Draw pictures of all 12 orbitals.arrow_forward
- Which of the following statements is true?a. All antibonding MOs are higher in energy than the atomic orbitals of which they arecomposed.b. Antibonding MOs have electron density mainly outside the space between the two nuclei.c. Electrons are never found in an antibonding MO.d. Both a. and b. are true.e. Both a. and c. are true.arrow_forwardIn at 6 p-orbital system with 6 p-π electrons, which MO is the HOMO? Is this MO symmetric or asymmetric? How many vertical nodes does it have? Circle a choice for each part.arrow_forwardThe bond in Cl2 can be explained by the end-on overlap between two p AOs, as shown inthis diagram. (a) Draw the bonding and antibonding MOs that would result from such an interaction. (b) What is the symmetry of each of these MOs?arrow_forward
- I am confused why this powerpoint slight says 'sideways overlap... leads to a p-bonding MO AND a p*-antibonding MO.' Isn't it one or the other? Why does it say AND? The orbitals can't have descrutive and constructive interaction at the same time right?arrow_forwardWhich MOs are the HOMO and the LUMO in the ground state? for the MOs of 1,3,5-hexatrienearrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Linear Combination of Atomic Orbitals LCAO; Author: Edmerls;https://www.youtube.com/watch?v=nq1zwrAIr4c;License: Standard YouTube License, CC-BY
Quantum Molecular Orbital Theory (PChem Lecture: LCAO and gerade ungerade orbitals); Author: Prof Melko;https://www.youtube.com/watch?v=l59CGEstSGU;License: Standard YouTube License, CC-BY