
Concept explainers
(a)
Interpretation:
The allylic carbons in the given compound are to be identified.
Concept introduction:
Hydrocarbons are formed by carbon and hydrogen atom. These atoms form a bond with each other and with other atoms through the sharing of electrons.
Answer to Problem 17.1P
The allylic carbon atoms in the given compound are shown below.
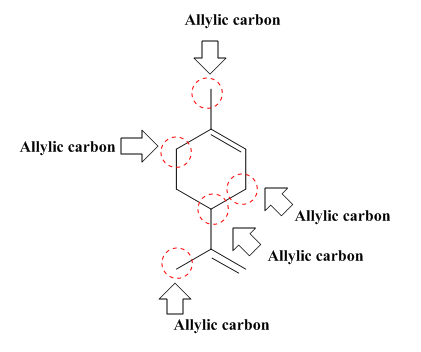
Explanation of Solution
The structure of a given compound is shown below.
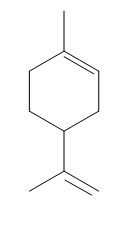
Figure 1
In Figure 1, there are five allylic carbon
Therefore, the allylic carbon atoms in the given compounds are shown below.

Figure 2
The allylic carbon atoms in the given compound are shown in Figure 2.
(b)
Interpretation:
The allylic carbons in the given compound are to be identified.
Concept introduction:
Hydrocarbons are formed by carbon and hydrogen atom. These atoms form a bond with each other and with other atoms through the sharing of electrons. Alkenes have carbon-carbon double bonds in them. The allylic carbon atoms are carbon atoms which are bonded to a carbon atom that make the double bond with another carbon atom.
Answer to Problem 17.1P
The allylic carbon atoms in the given compound are shown below.
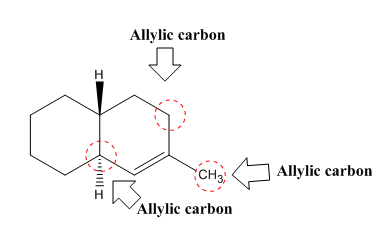
Explanation of Solution
The structure of a given compound is shown below.
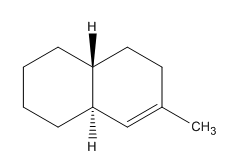
Figure 3
In Figure 3, there are three allylic carbon
Therefore, the allylic carbon atoms in the given compounds are shown below.

Figure 4
The allylic carbon atoms in the given compound are shown in Figure 4.
Want to see more full solutions like this?
Chapter 17 Solutions
EBK ORGANIC CHEMISTRY STUDY GUIDE AND S
- Provide the reagents and solvents (where appropriate) needed to bring about the following transformations. (a) CI (b)arrow_forwardWhat will be the color of the flame and the amount of soot if the following are ignited:(a) Hexane (b)Heptane(c) Cyclohexane (d) Cyclohexene (e) Benzene (f) Toluenearrow_forwardDraw the structures of the following compounds. (a) isobutyl bromidearrow_forward
- Describe how would you distinguish the following pairs, (a) Benzene and cyclohexane (b) Phenol and toluene (c) Phenol and benzoic acidarrow_forward11:43 Q1. (a) (c) (d) (b) Two stereoisomers of but-2-ene are formed when 2-bromobutane reacts with ethanolic potassium hydroxide. (i) Explain what is meant by the term stereoisomers. Library Name and outline a mechanism for the reaction of 2-bromo-2-methylpropane with ethanolic potassium hydroxide to form the alkene 2-methylpropene, (CH3)2C=CH₂ Name of mechanism Mechanism (ii) Draw the structures and give the names of the two stereoisomers of but-2-ene. Stereoisomer 1 Name (iii) Name this type of stereoisomerism. Select Name Stereoisomer 2 When 2-bromo-2-methylpropane reacts with aqueous potassium hydroxide, 2-methylpropan-2-ol is formed as shown by the following equation. CH3 H₂C-C-CH3 + KOH Br Page 2 of 14 CH3 H3C-C-CH3 + KBr ОН State the role of the hydroxide ions in this reaction. Write an equation for the reaction that occurs when CH3CH₂CH₂CH₂Br reacts with an excess of ammonia. Name the organic product of this reaction. Equation Name of product 9,284 Photos, 1,166 Videos For You…arrow_forwardConsider the tetracyclic compound with rings labeled A–D. (a) Which ring is the most reactive in electrophilic aromatic substitution? (b) Which ring is the least reactive in electrophilic aromatic substitution?arrow_forward
- 6. Describe concisely a chemical test to distinguish between the following pairs of compounds. (a) n-pentanol and 3-methylpentan-3-ol (b) Ethanal dan pentanal (c) Phenol and benzoic acidarrow_forwardF.33 Provide the IUPAC name for each of the following molecules. (a) (b) (c) (d) N. NH,arrow_forwardPredict the more stable bridge head alkene in the following compounds. (A) (B) (C) (D)arrow_forward
- (a) One test for the presence of an alkene is to add a smallamount of bromine, which is a red-brown liquid, and lookfor the disappearance of the red-brown color. This test doesnot work for detecting the presence of an aromatic hydrocarbon.Explain. (b) Write a series of reactions leading topara-bromoethylbenzene, beginning with benzene andusing other reagents as needed. What isomeric side productsmight also be formed?arrow_forward(a) Which is more reactive towards Electrophilic aromatic substitution (EAS)? (b) Which of the following is a a meta-substituted compound? (c) Which is the least reactive towards Electrophilic aromatic substitution (EAS)? (d) Which is an ortho-substituted compound?arrow_forwardProvide IUPAC names for the following compounds. (a) -OH (b)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





