
Concept explainers
(a)
Interpretation:
The detailed mechanism for the given reaction is to be drawn, and the major product is to be predicted.
Concept introduction:
Lithium aluminium hydride (LAH) or
The chemical behaviour of deuterium (
Answer to Problem 17.43P
The detailed mechanism for the given reaction is
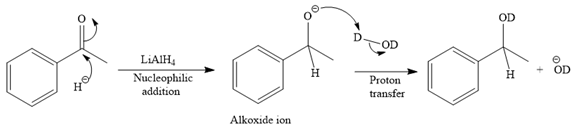
The major product of the given reaction:
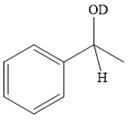
Explanation of Solution
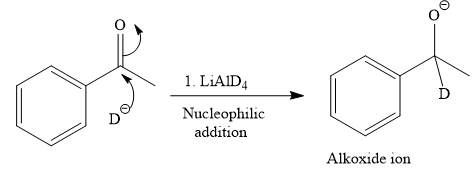
The given reaction is
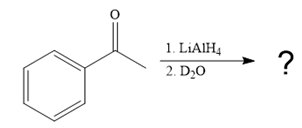
This alkoxide ion then attacks the deuterium (
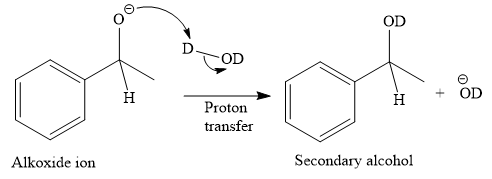
Thus, the final product of the given reaction is the secondary alcohol shown below:
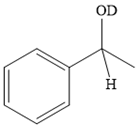
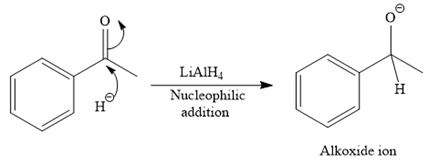
A ketone, when treated with a reducing agent such as Lithium aluminium hydride (LAH) or
(b)
Interpretation:
The detailed mechanism for the given reaction is to be drawn, and the major product is to be predicted.
Concept introduction:
Lithium aluminium hydride (LAH) or
The chemical behaviour of deuterium (
Answer to Problem 17.43P
The detailed mechanism for the given reaction is
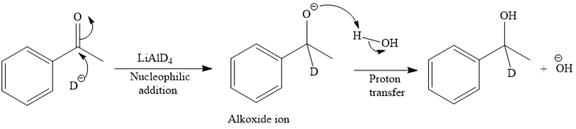
The major product of the given reaction:
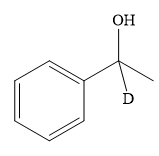
Explanation of Solution
The given reaction is
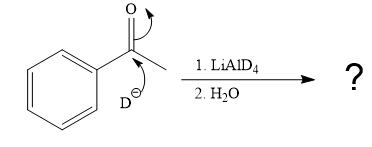
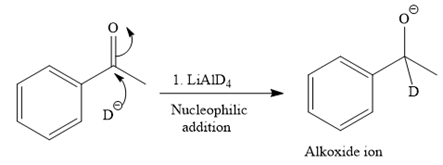
This alkoxide ion then attacks the proton (H) of water, which is the solvent used in the next step.
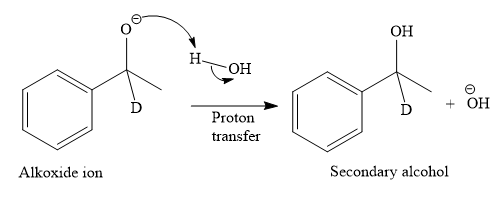
Thus, the final product of the given reaction is the secondary alcohol shown below:
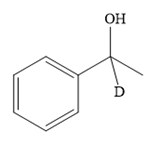
A ketone, when treated with a reducing agent such as Lithium aluminium hydride (LAH) or
(c)
Interpretation:
The detailed mechanism for the given reaction is to be drawn, and the major product is to be predicted.
Concept introduction:
Lithium aluminium hydride (LAH) or
The chemical behaviour of deuterium (
Answer to Problem 17.43P
The detailed mechanism for the given reaction is
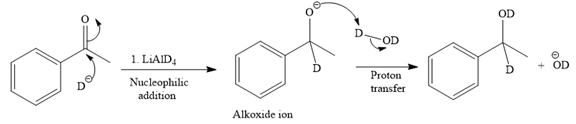
The major product of the given reaction:
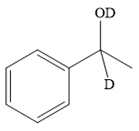
Explanation of Solution
The given reaction is
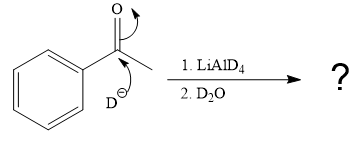
This alkoxide ion then attacks the deuterium (
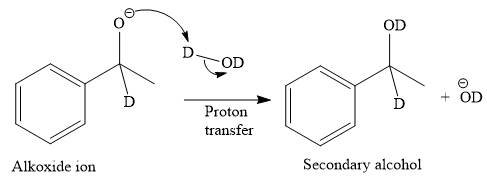
Thus, the final product of the given reaction is the secondary alcohol shown below:
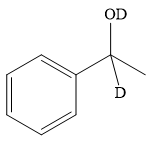
A ketone, when treated with a reducing agent such as Lithium aluminium hydride (LAH) or
Want to see more full solutions like this?
Chapter 17 Solutions
Organic Chemistry: Principles And Mechanisms: Study Guide/solutions Manual (second)
- Predict the major product(s) of each of the following reactions. Draw the complete, detailed mechanism that leads to the formation of each of those products. (b) (c) (a) HO;S HNO3 Cl2. ? H,SO4 ? FeCl3 AICI3 H3CO (d) (e) Br2, ? conc HNO3 ? FeBr3arrow_forwardDraw the complete, detailed E1 mechanism for each of the following reactions, and show all resonance structures, where applicable.arrow_forwardShown below is a two-step mechanism beginning with nucleophilic attack of water, and subsequent deprotonation with a base. Draw the arrows for the mechanism for both step 1 and step 2 and draw the intermediate product of in the box. + H-O OH + H₂Oarrow_forward
- When the following deuterium-labeled compound is treated with potassium tert-butoxide in DMF, a single product is observed. When the same substrate is heated in the presence of dilute potassium ethoxide in ethanol, a mixture of two products is formed. Provide the complete, detailed mechanism (curved arrows) for each reaction and label each reaction as E1 or E2. Note: deuterium is an isotope of hydrogen and can be treated similarly to hydrogen in chemical reactions but cannot be implied. D H KO/Bu DMF D H dilute KOET EtOH ?+ ?arrow_forwardPredict the product(s) and provide the complete mechanism for each reaction below.arrow_forward(b) While trying to synthesise product A following the reaction scheme shown below, the desired product was never obtained. Instead an unwanted product B was obtained in 100% yield. Provide the detailed mechanism of how this product formed and suggest a new synthesis, starting from the given starting material to obtain product A (No mechanism is required for the new synthesis). مفید مخافه میشود 1. Na OEt 2. Br 1. NaOEt B 2. ge -Br 100 % yield 1. NaOH Heat مبر 0% yieldarrow_forward
- When ethylbenzene reacts with bromine in a free radical halogenation, a small amount ofproduct that contains no bromine is formed. Show the mechanism of how the product isformed and the product. Explain why only a small amount of that product is formed.arrow_forwardDraw the complete, detailed mechanism (including curved arrows) for each of the following reactions occurring via (a) an E2 mechanism and (b) an E1 mechanism. If more than one possible product can be produced from the same type of mechanism, draw the complete mechanism that leads to each one. Pay attention to stereochemistry. (i) (ii) Br NaOH NaOH ? (iii) (iv) CI KOC(CH3)3 NaOCH3 H3C CH3 (v) CH3 (vi) CI CI КОН КОН ? 'CH3 "CH3arrow_forwardThe chemical behavior of deuterium (D or 2H) is essentially identical to that of hydrogen ('H). Therefore, D20 behaves the same as H,0, and CD3LI behaves the same as CH3LI. With this in mind, draw the detailed mechanism of each of the following reactions. Using the mechanism, predict the reaction products. (a) (b) (c) 1. CH3LI 1. CD3LI 1. CD3LI ? 2. D20 ? 2. H20 2. D20arrow_forward
- Predict the major products of the following reactions. Draw the complete, detailed mechanism that leads to the formation of each of those products.arrow_forwardProvide the mechanism for the following reaction. CI , AIC3arrow_forwardThe following isomers react separately with sodium hydroxide to give different products with the formulas shown. HO. .CI Но NaOH NaOH C3H1,02 (a) Draw the structure of each product. (b) Draw the mechanism that accounts for the formation of each of those products. (c) Explain why the isomeric reactants lead to different products.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

