
Concept explainers
Propose a structure consistent with each set of data.
a.
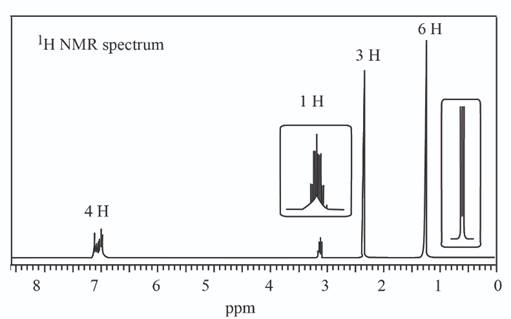
b.
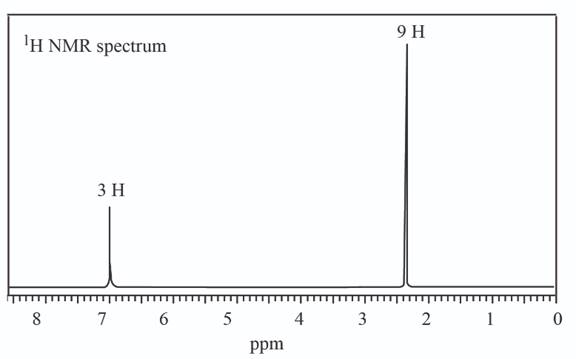
c.
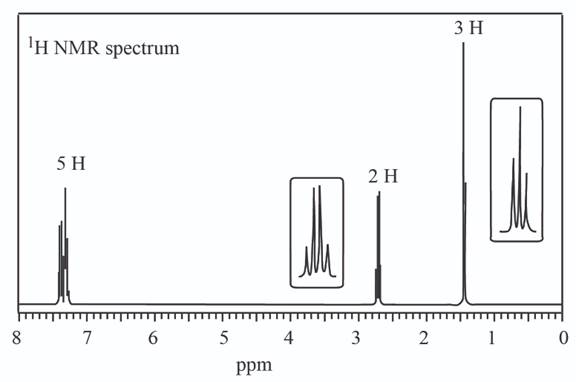
(a)
Interpretation: A structure consistent with the given set of data is to be predicted.
Concept introduction: Spectroscopy method is used to identify the structure of the molecule. It is based on the interactions between matter and electromagnetic radiations. Proton NMR spectroscopy identifies the number of hydrogen atoms present in a molecule and the nature of the functional group. The value of chemical peaks depends upon the chemical environment around the hydrogen atom.
Answer to Problem 17.55P
A structure consistent with the given set of data is shown below.
Explanation of Solution
The given set of
Information from
The
Information from
The observed chemical shift value at
The observed chemical shift value at
The observed chemical shift value at
The observed chemical shift value at
The possible structure of the compound based on the above analysis is,
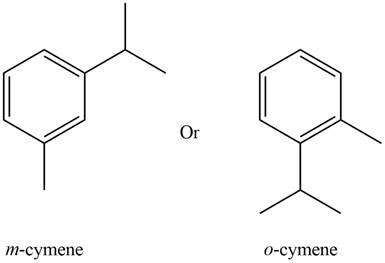
Figure 1
A structure consistent with the given set of data is shown in Figure 1.
(b)
Interpretation: A structure consistent with the given set of data is to be predicted.
Concept introduction: Spectroscopy method is used to identify the structure of the molecule. It is based on the interactions between matter and electromagnetic radiations. Proton NMR spectroscopy identifies the number of hydrogen atoms present in a molecule and the nature of the functional group. The value of chemical peaks depends upon the chemical environment around the hydrogen atom.
Answer to Problem 17.55P
A structure consistent with the given set of data is shown below.
Explanation of Solution
The given set of
Information from
Three signals are observed in
The
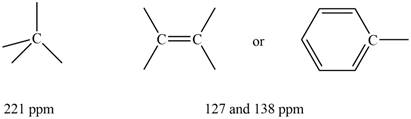
Figure 2
Information from
The observed chemical shift value at
The observed chemical shift value at
The possible structure of the compound based on the above analysis is,

Figure 3
A structure consistent with the given set of data is shown in Figure 3.
(c)
Interpretation: A structure consistent with the given set of data is to be predicted.
Concept introduction: Spectroscopy method is used to identify the structure of the molecule. It is based on the interactions between matter and electromagnetic radiations. Proton NMR spectroscopy identifies the number of hydrogen atoms present in a molecule and the nature of the functional group. The value of chemical peaks depends upon the chemical environment around the hydrogen atom.
Answer to Problem 17.55P
A structure consistent with the given set of data is shown below.
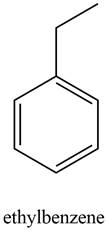
Explanation of Solution
The given set of
Information from IR data:
The
Information from
The observed chemical shift value at
The observed chemical shift value at
The observed chemical shift value at
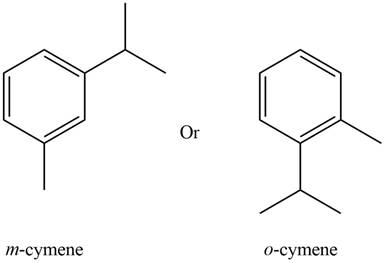
The possible structure of the compound based on the above analysis is,
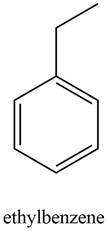
Figure 4
A structure consistent with the given set of data is shown in Figure 4.
Want to see more full solutions like this?
Chapter 17 Solutions
ORG CHEM LL W/ LL SG&CONPLUS PKG>IC<
- 3 The UV−vis spectrum for K[VO(O2)Hheida] shows an absorption at 435 nm (ε = 4 × 102 M−1 cm−1). What phenomenon is presumed to be responsible for this absorption?arrow_forwardA hydrocarbon, compound B, has molecular formula C6H6, and gave an NMR spectrum with two signals: delta 6.55 pm and delta 3.84 pm with peak ratio of 2:1. When warmed in pyridine for three hr, compound B quantitatively converts to benzene. Mild hydrogenation of B yielded another compound C with mass spectrum of m/z 82. Infrared spectrum showed no double bonds; NMR spectrum showed one broad peak at delta 2.34 ppm. With this information, address the following questions. a) How many rings are in compound C? b) How many rings are probably in B? How many double bonds are in B? c) Can you suggest a structure for compounds B and C? d) In the NMR spectrum of B, the up-field signal was a quintet, and the down field signal was a triplet. How must you account for these splitting patterns?arrow_forwardThe NMR spectra for compound 1 were acquired in a 7.5 mg / 0.6 mL solution ofCDCl3 . The 1H and 13C peaks are also listedbelow. Provide a full analysis of the NMR spectra for compound 1. correct assignment of NMR spectra of both 13C spectra. correct rationalisation of 13C spectrum1H NMR (400 MHz, CDCl3) δ 7.73 (d, J = 9.5 Hz, 1H), 7.56 (ddd, J = 8.5, 7.5, 1.6 Hz, 1H),7.51 (dd, J = 7.5, 1.6 Hz, 1H), 7.36 (d, J = 8.5 Hz, 1H), 7.30 (dd, J = 8.5, 7.5 Hz, 1H), 6.45(d, J = 9.5 Hz, 1H).13C NMR (101 MHz, CDCl3) δ 160.79, 154.09, 143.43, 131.85, 127.87, 124.44, 118.86,116.94, 116.74.Note: There are two carbon peaks in the 13C spectrum that are so close together that they are not differentiable at the resolution in this experiment. you should be able to assign these peaks to one of two carbon atoms in 1.arrow_forward
- For each compound shown below,(1) sketch the 13C NMR spectrum (totally decoupled, with a singlet for each type of carbon), showing approximatechemical shifts.(2) show the multiplicity expected for each signal in the off-resonance-decoupled spectrum.(3) sketch the spectra expected using the DEPT-90 and DEPT-135 techniques. 3-chloropropenearrow_forwardA compound shows strong, very broad IR absorption in the region 3300–3600 cm-1 and strong, sharp absorption at 1715 cm-1. What functional group accounts for both of these absorptions?arrow_forwardChoose the structure corresponding to the given 1H and 13C NMR spectraarrow_forward
- Reaction of butanenitrile (CH3CH2CH2CN) with methylmagnesium bromide (CH3MgBr), followed by treatment with aqueous acid, forms compound G. G has a molecular ion in its mass spectrum at m/z = 86 and a base peak at m/z = 43. G exhibits a strong absorption in its IR spectrum at 1721 cm−1 and has the 1H NMR spectrum given below. What is the structure of G? We will learn about the details of this reaction in Chapter 22.arrow_forwardExplain the order of chemical shifts of the carbon atoms in the 13C spectra of cyclohexanone (fig 22.6 ) and adipic acid (fig .22.9) attached below .arrow_forwardPropose a structure consistent with following set of spectral data: C3H6Br2: IR peak at 3000–2850 cm−1; NMR (ppm): 2.4 (quintet) 3.5 (triplet)arrow_forward
- Draw a structure consistent with the following set of data: a hydrocarbon with a molecular ion at m/z = 68 and IR absorptions at 3310, 3000−2850, and 2120cm−1.arrow_forward1) what is the struture of B, mechanism for its formation and assigment of the IR and 13C spectra 2) what is the struture of C, mechanism for its formation and assigment of the 1H NMR spectra 3) what is the mechanism of C to D in step 4arrow_forwardAn unknown compound C3H2NCl shows moderately strong IR absorptions around 1650 cm-1 and 2200 cm-1. Its NMR spectrum consists of two doublets (J = 14 Hz) at δ 5.9 and δ 7.1. Propose a structure consistent with this data?arrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
