
ORGANIC CHEMISTRY (LOOSELEAF) >CUSTOM<
5th Edition
ISBN: 9781259995941
Author: SMITH
Publisher: MCG CUSTOM
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 17.60P
Rizatriptan (trade name Maxalt) is a prescription drug used for the treatment of migraines. (a) How many aromatic rings does rizatriptan contain? (b) Determine the hybridization of each
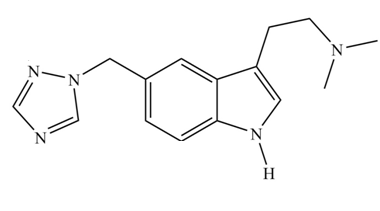
rizatriptan
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Stanozolol is an anabolic steroid that promotes muscle growth. Althoughstanozolol has been used by athletes and body builders, many physicaland psychological problems result from prolonged use and it is bannedin competitive sports.
Draw all reasonable resonance structures for stanozolol.
Rizatriptan (trade name Maxalt) is a prescription drug used for the treatment of migraines. (a) How many aromatic rings does rizatriptan contain? (b) Determine the hybridization of each N atom. (c) In what type of orbital does the lone pair on each N reside? (d) Draw all the resonance structures for rizatriptan that contain only neutral atoms. (e) Draw all reasonable resonance structures for the five-membered ring that contains three N atoms.
Rank the labeled bonds in santalbic acid, a fatty acid obtained from the seeds of the sandalwood tree used in cosmetics, in order of increasing bond length.
Chapter 17 Solutions
ORGANIC CHEMISTRY (LOOSELEAF) >CUSTOM<
Ch. 17 - Prob. 17.1PCh. 17 - Problem 17.2 What orbitals are used to form the...Ch. 17 - Problem-17.3. Give the IUPAC name for each...Ch. 17 - Prob. 17.4PCh. 17 - Problem-17.5 What is the structure of propofol,...Ch. 17 - Problem 17.6 What is the structure of a compound...Ch. 17 - Problem 17.7 How many NMR signals does each...Ch. 17 - Prob. 17.8PCh. 17 - Prob. 17.9PCh. 17 - Prob. 17.10P
Ch. 17 - Prob. 17.11PCh. 17 - Prob. 17.12PCh. 17 - Prob. 17.13PCh. 17 - Problem 17.14 Januvia, the trade name for...Ch. 17 - Prob. 17.15PCh. 17 - Problem 17.16 Rank the following compounds in...Ch. 17 - Problem 17.17 Draw the seven resonance structures...Ch. 17 - Prob. 17.18PCh. 17 - Prob. 17.19PCh. 17 - Prob. 17.20PCh. 17 - Prob. 17.21PCh. 17 - Problem 17.22 How many NMR signals does ...Ch. 17 - 17.23 Name each compound and state how many lines...Ch. 17 - Prob. 17.24PCh. 17 - Prob. 17.25PCh. 17 - Prob. 17.26PCh. 17 - 17.27 Give the IUPAC name for each compounds.
a....Ch. 17 - 17.28 Draw a structure corresponding to each...Ch. 17 - 17.29 a. Draw the 14 constitutional isomers of...Ch. 17 - Prob. 17.30PCh. 17 - Prob. 17.31PCh. 17 - Prob. 17.32PCh. 17 - 17.33 Label each compound as aromatic,...Ch. 17 - Prob. 17.34PCh. 17 - 17.35 Pentalene, azulene, and heptalene are...Ch. 17 - 17.36 The purine heterocycle occurs commonly in...Ch. 17 - Prob. 17.37PCh. 17 - 17.38
How many electrons does C contain?
How...Ch. 17 - Prob. 17.39PCh. 17 - 17.40 Explain the observed rate of reactivity of...Ch. 17 - 17.41 Draw a stepwise mechanism for the following...Ch. 17 - Prob. 17.42PCh. 17 - 17.43 Draw additional resonance structures for...Ch. 17 - Prob. 17.44PCh. 17 - Prob. 17.45PCh. 17 - 17.46 Which compound in each pair is the stronger...Ch. 17 - 17.47 Treatment of indene with forms its...Ch. 17 - Prob. 17.48PCh. 17 - 17.49 Draw the conjugate bases of pyrrole and...Ch. 17 - 17.50 a. Explain why protonation of pyrrole occurs...Ch. 17 - Prob. 17.51PCh. 17 - Prob. 17.52PCh. 17 - 17.53 How many signals does each compound...Ch. 17 - 17.54 Which of the diethylbenzene isomers (ortho,...Ch. 17 - 17.55 Propose a structure consistent with each...Ch. 17 - 17.56 Propose a structure consistent with each...Ch. 17 - 17.57 Thymol (molecular formula ) is the major...Ch. 17 - 17.58 You have a sample of a compound of molecular...Ch. 17 - 17.59 Explain why tetrahydrofuran has a higher...Ch. 17 - 17.60 Rizatriptan (trade name Maxalt) is a...Ch. 17 - 17.61 Zolpidem (trade name Ambien) promotes the...Ch. 17 - 17.62 Answer the following questions about...Ch. 17 - 17.63 Stanozolol is an anabolic steroid that...Ch. 17 - Prob. 17.64PCh. 17 - 17.65 Use the observed data to decide whether C...Ch. 17 - Prob. 17.66PCh. 17 - Prob. 17.67PCh. 17 - Prob. 17.68PCh. 17 - 17.69 Although benzene itself absorbs at in its ...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- As mentioned in Problem 3-53, the statin drugs, such as simvastatin (Zocor), pravastatin (Pravachol), and atorvastatin (Lipitor) are the most widely prescribed drugs in the world. (a) Are the two indicated bonds on simvastatin cis or trans? (b) What are the cis/trans relationships among the three indicated bonds on pravastatin? (c) Why cant the three indicated bonds on atorvastatin be identified as cis or trans?arrow_forwardAm I drawing the resonance correctly for each? Thank youarrow_forwardare these aromatic, antiaromatic, or nonaromatic? Draw in all lonepairs and indicate the orbital each lone pair resides in.arrow_forward
- Hydrocarbon A possesses a significant dipole, even though it iscomposed of only C—C and C—H bonds. Explain why the dipole arisesand use resonance structures to illustrate the direction of the dipole.Which ring is more electron rich?arrow_forwardAnswer the following questions about erlotinib and terbinafine. Erlotinib,sold under the trade name Tarceva, was introduced in 2004 for thetreatment of lung cancer. Terbinafine is an antifungal medication used totreat ringworm and fungal nail infections. Rank the labeled bonds in terbinafine in order of increasing bondstrength.arrow_forwardWhich line (A or B) corresponds to the energy change for an ortho/para substitution?arrow_forward
- Draw all possible structural (constitutional and cis-trans) isomers in bond-line presentations for the following compounds: A) C4H9Br (4 isomers) B) C3H7N (non cyclic compounds only) (at least 8 isomers including stereoisomers). C) C2H4F2 (not that many possibilities) D) C4H10O (7 isomers) E) C3H6O (compounds with single bonds only) (3 isomers) F) F2CCH2 (pay attention that the connectivity in this compound is already defined) G) C2H2Br2 (3 isomers including stereoisomers)arrow_forwardThe following compounds a) are NOT resonance structures, they aredifferent compounds. b) The following compound is aromaticarrow_forwardThapsigargin is a natural product with promising anticancer properties.a.At which sites can thapsigargin hydrogen bond to another molecule like itself? b.At which sites can thapsigargin hydrogen bond to water? c.How many sp2 hybridized C's are present? d.How many sp3 hybridized 3° C's are present?arrow_forward
- Draw ALL REASONABLE resonance structures and the resonance hybrids of the compounds below.arrow_forward1. Draw an aromatic ring, which contains only hydrogens and 5 carbons; add charges and lonepairs if needed 2. Draw a non-aromatic ring which contains only hydrogen and 5 carbons and have conjugateddouble bonds; add charges and lone pairs if needed.arrow_forwardThe calicene molecule has a different dipolar moment of 0 and also thedipole points to the larger ring. Draw a structure resonance where you can see that distribution of loads and using the concept of aromaticity and resonance structures, explain why the moment dipolarit's different from zeroarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Types of bonds; Author: Edspira;https://www.youtube.com/watch?v=Jj0V01Arebk;License: Standard YouTube License, CC-BY