
Concept explainers
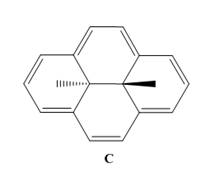
Use the observed
aromatic. C shows
signals at
extent?
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Organic Chemistry-Package(Custom)
- How many 1H NMR signals does each alkene exhibit?arrow_forwardAssign the protons to the appropriate 1H NMR signalarrow_forwardBased on the structures given below, which of the following statements is entirely true? Quinoline NH Isoquinoline Indole Isoindole All four compounds are non-aromatic, with 12 pi-electrons each and are equal in basicity. Only quinoline and Isoquinoline having 10 pi-electrons each are aromatic while indole and isoindole are antiaromatic having 8 pi-electrons each The nitrogen atoms of quinoline and isoquinoline are sp2 hybridized while the nitrogen atoms of indole and isoindole are sp³ hybridized. All four compounds are aromatic, with 10 pi-electrons each and are equal in basicity. All four compounds are aromatic, with 10 pi-electrons each, and with quinoline and isoquinoline being stronger bases than indole and isoindarrow_forward
- Write out the structure of the products and predict an NMR spectrum from each one.arrow_forwardThe 'H NMR spectrum of compound A (C3H100) has four signals: a multiplet at 8 = 7.25-7.32 ppm (5 H), a singlet at d = 5.17 ppm (1 H), a quartet at d = 4.98 ppm (1 H), and a doublet at ô = 1.49 ppm (3 H). There are 6 signals in its 13C NMR spectrum. The IR spectrum has a broad absorption in the -3200 cm-1 region. Compound A reacts with KMNO4 in a basic solution followed by acidification to give compound B with the molecular formula C7H6O2. Draw structures for compounds A and B.arrow_forwardWhich of the diethylbenzene isomers (ortho, meta, or para) corresponds to each set of 13C NMR spectral data?[A] 13C NMR signals: 16, 29, 125, 127.5, 128.4, and 144 ppm[B] 13C NMR signals: 15, 26, 126, 128, and 142 ppm[C] 13C NMR signals: 16, 29, 128, and 141 ppmarrow_forward
- Assign the resonances in this C-NMR spectrum.arrow_forwardExplain why aromatic protons resonate downfield and aliphatic resonate upfield.arrow_forwardExplain why the carbonyl carbon of an aldehyde or ketone absorbs farther downeld than the carbonyl carbon of an ester in a 13C NMR spectrum.arrow_forward
- Which set of underlined hydrogens has its 1H NMR signal at a higher frequency?arrow_forwardWhen compound A (C5H12O) is treated with HBr, it forms compound B (C5H11Br). The 1H NMR spectrum of compound A has a 1H singlet, a 3Hdoublet, a 6H doublet, and two 1H multiplets. The 1H NMR spectrum of compound B has a 6H singlet, a 3H triplet, and a 2H quartet. Identifycompounds A and B.arrow_forwardBased on the structures given below, which of the following statements is entirely true? N Quinoline NH Isoquinoline Indole Isoindole All four compounds are non-aromatic, with 12 pi-electrons each and are equal in basicity. All four compounds are aromatic, with 10 pi-electrons each and are equal in basicity. Only quinoline and isoquinoline having 10 pi-electrons each are aromatic while indole and isoindole are antiaromatic having 8 pi-electrons each. All four compounds are aromatic, with 10 pi-electrons each, and with quinoline and isoquinoline being stronger bases than indole and isoindole. The nitrogen atoms of quinoline and isoquinoline are sp2 hybridized while the nitrogen atoms of indole and isoindole are sp³ hybridized Previousarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





