
(a)
Interpretation:
The structure of six-membered cyclic hemiacetal formed by 5-hydroxyhexanal should be drawn.
Concept Introduction:
Hemiacetal is a molecule formed by the addition of an alcohol to an
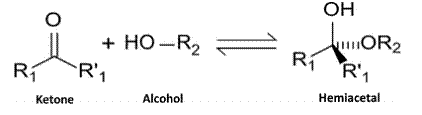
Answer to Problem 17.71P
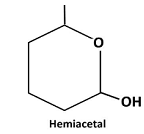
Explanation of Solution
Reaction of the aldehyde group and the hydroxyl group forms a six-membered cyclic hemiacetal.
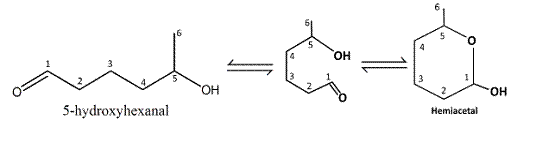
Therefore, the structure hemiacetal formed from 5-hydroxyhexanalis as follows:
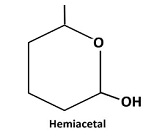
(b)
Interpretation:
The number of possible stereoisomers of 5-hydroxyhexanal should be determined.
Concept Introduction:
Stereoisomers are the compounds that are differ only in the spatial arrangements of their atoms. For a molecule with n stereoisomers, a maximum of
Answer to Problem 17.71P
5-hydroxyhexanal has 2 stereoisomers.
Explanation of Solution
For a molecule with n stereoisomers, a maximum of
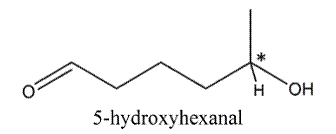
In 5-hydroxyhexanal, there is one stereocenter.
Therefore, number of stereoisomers can be calculated as follows:
Thus, number of stereoisomers will be 2.
(c)
Interpretation:
The number of possible stereoisomers of hemiacetal of 5-hydroxyhexanal should be calculated.
Concept Introduction:
Stereoisomers are the compounds that are differ only in the spatial arrangements of their atoms. For a molecule with n stereoisomers, a maximum of
Answer to Problem 17.71P
Hemiacetal of 5-hydroxyhexanal has 4 stereoisomers.
Explanation of Solution
For a molecule with n stereoisomers, a maximum of
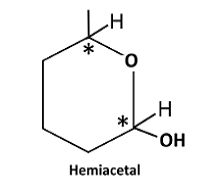
In hemiacetal of 5-hydroxyhexanal, there are two stereo centres.
Therefore, number of stereoisomers can be calculated as follows:
Thus, number of stereoisomers are 4.
Want to see more full solutions like this?
Chapter 17 Solutions
Bundle: Introduction To General, Organic And Biochemistry, 11th + Owlv2, 1 Term (6 Months) Printed Access Card
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l)CH3CH2CH2CO2H(l)+CH2CH3OH(l)⟶H+CH3CH2CH2CO2CH2CH3(l)+H2O(l) A chemist ran the reaction and obtained 5.40 g of ethyl butyrate. What was the percent yield, The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.45g of butanoic acid and excess ethanol?arrow_forward18-28 Arrange these compounds in order of increasing acidity: benzoic acid, benzyl alcohol, phenol.arrow_forward18-19 The following compounds have approximately the same molecular weight: hexanoic acid, heptanal, and 1-heptanol. Arrange them in order of increasing boiling point.arrow_forward
- 17-47 What is the characteristic structural feature of a hemiacetal? Of an acetal?arrow_forward18-29 Complete the equations for these acid—base reactions. (a) (b) (c) (d) (e)arrow_forward18-41 Complete these examples of Fischer esterification. In each case, assume an excess of the alcohol.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


