
Chemistry: Central Science (Looseleaf)-Package
13th Edition
ISBN: 9780133943566
Author: Brown
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 17E
The drawing below shows the overlap of two hybrid orbitals to form a bond in a hydrocarbon.
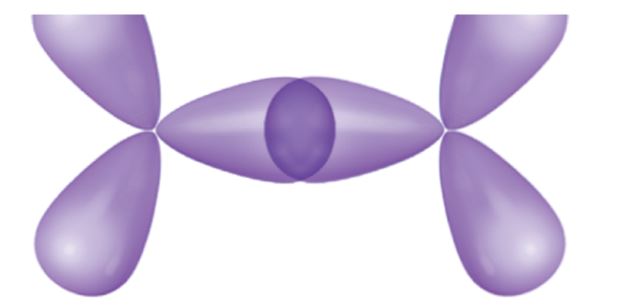
ii. C-Cw, or
iii. C-C?
b. Which of the following could be the identity of the hydrocarbon:
i.CH 4
ii.C 2H 6, or
iii.C 2H 4
iv.C 2H 2? (Section 9.6)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
One of the first drugs to be approved for use in treatment of acccquired immune deficiency syndrome (AIDS) was azidothymidine (AZT). Complete the Lewis structure for AZT
a. How many carbon atoms are sp3 hybridised?
b. How many carbon atoms are sp2 hybridised?
c. Which atom is sp hybridised?
d. How many σ bonds are in the molecule?
e. How many π bonds are in the molecule?
f. What is the N9N9N bond angle in the azide (--N3) group?
g. What is the H--Q--C bond angle in the side group attached to the five-membered ring?
h. What is the hybridization of the oxygen atom in the --CH2OH group?
A certain AB4 molecule has a square-planar molecular geometry.
Which of the following statements about the molecule is or are true?
1. The molecule has four electron domains about the central atom A.
II. The B-A-B angles between neighboring B atoms is 90°.
IlI. The molecule has two nonbonding pairs of electrons on atom A.
A. Only one of the statements is true
B. Statements I and Il are true
C. Statements I and lll are true
D. Statements Il and IIl are true
E. All three statements are true
Chapter 17 Solutions
Chemistry: Central Science (Looseleaf)-Package
Ch. 17.1 - Calculate the formal charge on the indicated atom...Ch. 17.1 - The hypochlorite ion, CIO- , is the active...Ch. 17.1 - Prob. 17.2.1PECh. 17.1 - a. Triazine, C3 H3N3, is like benzene except that...Ch. 17.2 - Prob. 17.3.1PECh. 17.2 - Prob. 17.3.2PECh. 17.2 - Prob. 17.4.1PECh. 17.2 - Prob. 17.4.2PECh. 17.2 - Prob. 17.5.1PECh. 17.2 - Prob. 17.5.2PE
Ch. 17.2 - Prob. 17.6.1PECh. 17.2 - Prob. 17.6.2PECh. 17.3 - Prob. 17.7.1PECh. 17.3 -
8.103 The compound chloral hydrate, known in...Ch. 17.3 - Barium azide is 62.04% Ba and 37.96% N. Each azide...Ch. 17.3 - Acetylene (C2H2) and nitrogen (N2) both contain a...Ch. 17.3 - Prob. 17.9.1PECh. 17.3 - Prob. 17.9.2PECh. 17.4 - Prob. 17.10.1PECh. 17.4 - Prob. 17.10.2PECh. 17.4 - Prob. 17.11.1PECh. 17.4 - A new compound is made that has a C-C bond length...Ch. 17.4 - A new compound is made that has an N-N bond length...Ch. 17.4 - Prob. 17.12.2PECh. 17.5 - Prob. 17.13.1PECh. 17.5 - An ionic substance of formula MX has a lattice...Ch. 17.5 - Prob. 17.14.1PECh. 17.5 - Prob. 17.14.2PECh. 17.5 - Prob. 17.15.1PECh. 17.5 - Consider the collection of nonmetallic elements 0,...Ch. 17.6 - The substance chlorine monoxide, CIO(g), is...Ch. 17.6 -
[8.87]
a. using the electronegativities of Br...Ch. 17.6 - Prob. 17.17.1PECh. 17.6 - Although I3- is a known ion, F3- is not. a. Draw...Ch. 17 - Prob. 1DECh. 17 -
9.13
a. An AB2 molecule is linear. How...Ch. 17 - Give the electron-domain and molecular geometries...Ch. 17 - Prob. 3ECh. 17 - Prob. 4ECh. 17 - Prob. 5ECh. 17 - Prob. 6ECh. 17 - Prob. 7ECh. 17 - Prob. 8ECh. 17 - Azo dyes are organic dyes that are used for many...Ch. 17 - Prob. 10ECh. 17 - 9.1 A certain AB4, molecule has a "seesaw" shape...Ch. 17 - Prob. 12ECh. 17 - Prob. 13ECh. 17 - Prob. 14ECh. 17 - Prob. 15ECh. 17 - In the hydrocarbon a. What is the hybridization at...Ch. 17 - The drawing below shows the overlap of two hybrid...Ch. 17 - Prob. 18ECh. 17 -
9.10 The following is part of a molecular...Ch. 17 - a. Methane (CH4) and the perchlorate ion (C104-)...Ch. 17 - Prob. 21ECh. 17 - Prob. 22ECh. 17 - Prob. 23ECh. 17 - Prob. 24ECh. 17 - In which of these molecules or ions does the...Ch. 17 - Prob. 26ECh. 17 - How many nonbonding electron pairs are there in...Ch. 17 - Prob. 28ECh. 17 - Prob. 29ECh. 17 - Prob. 30ECh. 17 - Prob. 31ECh. 17 - Prob. 32ECh. 17 - Prob. 33ECh. 17 - Prob. 34ECh. 17 - Ammonia, NH3 reacts with incredibly strong bases...Ch. 17 - In which of the following AFn molecules or ions is...Ch. 17 - a. Explain why BrF4 is square planar, whereas...Ch. 17 -
9.34 Name the proper three-dimensional molecule...Ch. 17 - Prob. 39ECh. 17 - Prob. 40ECh. 17 - a. (a) Is the molecule BF3 polar or nonpolar? b....Ch. 17 - Prob. 42ECh. 17 - Predict whether each of the following molecules is...Ch. 17 - Prob. 44ECh. 17 - Prob. 45ECh. 17 - Prob. 46ECh. 17 - For each statement, irldicate whether it is true...Ch. 17 - Draw sketches illustrating the overlap between the...Ch. 17 - For each statement, indicate whether it is true or...Ch. 17 - Consider the SC12 molecule. a. What IS the...Ch. 17 - Prob. 51ECh. 17 - Prob. 52ECh. 17 - Prob. 53ECh. 17 - Prob. 54ECh. 17 - Prob. 55ECh. 17 - Prob. 56ECh. 17 - a. Draw Lewis structures for ethane (C2He),...Ch. 17 - a. Draw Lewis structures for ethane (C2He),...Ch. 17 - Prob. 59ECh. 17 - Prob. 60ECh. 17 - Prob. 61ECh. 17 - Prob. 62ECh. 17 - In the formate ion, HC02- , the carbon atom is the...Ch. 17 - Prob. 64ECh. 17 - Prob. 65ECh. 17 - Prob. 66ECh. 17 - Prob. 67ECh. 17 - a. If you combine two atomic orbitals on two...Ch. 17 - Prob. 69ECh. 17 - Indicate whether each statement is true or false....Ch. 17 - Prob. 71ECh. 17 - Prob. 72ECh. 17 - Prob. 73ECh. 17 - Prob. 74ECh. 17 - Prob. 75ECh. 17 - Prob. 76ECh. 17 - Determine the electron configurations for CN+, CN,...Ch. 17 - Prob. 78ECh. 17 - Consider the molecular orbitals of the P2...Ch. 17 - Prob. 80ECh. 17 - Consider the following XF4 ions: PF4, BrF4-,...Ch. 17 -
9.88 Consider the molecule PF4Cl....Ch. 17 - Prob. 83AECh. 17 - Fill in the blank spaces in the following chart....Ch. 17 - Prob. 85AECh. 17 - Prob. 86AECh. 17 - Prob. 87AECh. 17 - Prob. 88AECh. 17 - Prob. 89AECh. 17 - Prob. 90AECh. 17 - Prob. 91AECh. 17 - Prob. 92AECh. 17 - In ozone, 03, the two oxygen atoms on the ends Of...Ch. 17 - Butadiene, C4H6, is a planar molecule that has the...Ch. 17 - The structure of borazine, B3N3H6, is a...Ch. 17 - Prob. 96AECh. 17 - Prob. 97AECh. 17 - Prob. 98AECh. 17 - Prob. 99AECh. 17 - Prob. 100AECh. 17 - Prob. 101AECh. 17 - Consider the following AB3 molecules and ions-...Ch. 17 - Prob. 103AECh. 17 - Prob. 104AECh. 17 - Prob. 105AECh. 17 - Prob. 106AECh. 17 - Prob. 107AECh. 17 - Prob. 108AECh. 17 - Determine whether the following molecules are...Ch. 17 - Prob. 110IECh. 17 - Prob. 111IECh. 17 - Prob. 112IECh. 17 - Prob. 113IECh. 17 - Prob. 114IECh. 17 - Prob. 115IECh. 17 - Prob. 116IECh. 17 - Prob. 117IECh. 17 - Prob. 118IECh. 17 - Prob. 119IECh. 17 - Prob. 120IE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A compound of chlorine and fluorine. ClFn, reacts at about 75C with uranium metal to produce uranium hexafluoride, UF6, and chlorine monofluoridc. ClF(g). A quantity of uranium produced 7.04 g UF6, and 343 mL ClF at 75C and 2.50 atm. What is the formula (n) of the compound? Predict the molecular geometry using the VSEPR model.arrow_forwardFor each of the following molecules, state the bond angle (or bond angles, as appropriate) that you would expect to see on the central atom based on the simple VSEPR model. Would you expect the actual bond angles to be greater or less than this? a CCl4 b SCl2 c COCl2 d AsH3arrow_forwardThe antibiotic thiarubin-A was discovered by studying the feeding habits of wild chimpanzees in Tanzania. The structure for thiarubin-A is a. Complete the Lewis structure, showing all lone pairs of electrons. b. Indicate the hybrid orbitals used by the carbon and sulfur atoms in thiarubin-A. c. How many and bonds are present in this molecule?arrow_forward
- Allene (1,2-propadicne), a gas, has the following structure: What is the hybridization at each carbon atom? What is the geometry about each carbon atom? Using valencebond theory, describe the bonding about the center carbon atom. 1,3-dichloroallene is a derivative of allene with the following structure: Is this a polar molecule? (If you have difficulty visualizing the structure of this molecule, you might build a molecular model of it.)arrow_forwardA model of the organic compound allene is shown below. (a) Explain why the allene molecule is not flat. That is. explain why the CH2 groups at opposite ends do not lie in the same plane. (b) What is the hybridization of each of the carbon atoms in allene? (c) What orbitals overlap to form the bonds between carbon atoms in allene?arrow_forward7.85 Consider the molecule whose structure is shown below. (a) How many carbon atoms are sp3 hybridized? (b) What is the shape of the molecule around the carbon atom marked A? (c) What is the shape of the molecule around the carbon atom marked B?arrow_forward
- Consider acetyl salicylic acid, better known as aspirin. Its structure is (a) How many sigma and pi bonds are there in aspirin? (b) What are the approximate values of the angles marked (in blue) A, B, and C? (c) What is the hybridization of each atom marked (in red) 1, 2, and 3?arrow_forwardIdentify the molecules with a dipole moment: (a) SF4 (b) CF4 (c) Cl2CCBr2 (d) CH3Cl (e) H2COarrow_forwardWhat are the bond angles predicted by the VSEPR model about the carbon atom in the formate ion, HCO2? Considering that the bonds to this atom are not identical, would you expect the experimental values to agree precisely with the VSEPR values? How might they differ?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY