
Concept explainers
Give stereochemical formulas for compounds A–Q:
(a)
(b)
(c)
(d)
(e)
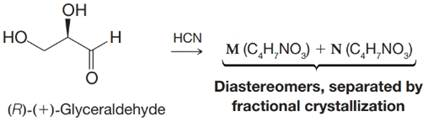
(f)
(g)
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Organic Chemistry, 12e Study Guide/Student Solutions Manual
Additional Science Textbook Solutions
CHEMISTRY-TEXT
Elementary Principles of Chemical Processes, Binder Ready Version
Organic Chemistry (8th Edition)
Introductory Chemistry (5th Edition) (Standalone Book)
Chemistry: The Molecular Nature of Matter
- 2) Starting with Benzene how can you prepare the following: H,C EN —ОН HO ,S NH,arrow_forwardDraw the product for the following reactionsarrow_forwardCompound A, C 10H 18O, undergoes reaction with dilute H 2SO 4 at 50 °C to yield a mixture of two alkenes, C 10H 16. The major alkene B, gives only cyclopentanone after ozone treatment followed by reduction with zinc in acetic acid. Which of the following reactions are correct. Can be more than one answerarrow_forward
- Predict the products of the following acid-base reactions. If the equilibrium would not result in the formation of appreciable amounts of products, you should so indicate. In each case label the stronger acid, the stronger base, the weaker acid, and the weaker base: (a) CH3CH=CH2 + NANH2 (d) CH3C=C: + CH;CH2OH → (e) CH3C=C:- + NH¾CI – | (b) CH;C=CH + NaNH2 (c) CH3CH2CH3 + NANH2 → | HASarrow_forwardThe acid-catalyzed hydrolysis of an ester converts an ester into a carboxylic acid. Although there are two O atoms that can be protonated, the first step in the mechanism is believed to be protonation of the oxygen in the C=0 group. Based on charge stability, why is it favorable to protonate that oxygen? Hint: Draw out the products of each protonation. + Hо HO, НО Carboxylic acid Ester Alcoholarrow_forwardIndicate the stereochemistry of the product of SN2 reaction: NaOH Br CH3 CH;OH Name the product:arrow_forward
- Identify the functional groups in the following molecules. (Use names from the table below. List each class of functional group only once. If there are fewer than 3 functional groups, leave an appropriate number of answer boxes empty.) a) b) N- -CH3 Name Structure Alkene Arene Structures of Some Common Functional Groups Alkyne -C=C1 Halide ** Nitro Thiol Name Aldehyde Ketone Structure DO SH 0-0² 'H 0=0arrow_forwardIdentify the following reactions as either SN1, SN2, E1, or E2: (a) Br CHCH3 CH=CH2 кон (b) Br OCH3 .CHCH3 .CHCH3 CH3OH Heatarrow_forwardIdentify the functional groups in the following molecules. (Use names from the table below. List each class of functional group only once. If there are fewer than 3 functional groups, leave an appropriate number of answer boxes empty.) CH3 a) b) H3C Alkene Name Structure H Arene Structures of Some Common Functional Groups Alkyne —C=C1 Halide Nitro Thiol Name Aldehyde Ketone Structure SH ASH 0-0²- H 010arrow_forward
- Draw the structural formula of compounds Q,R,T,U and Warrow_forwardPropose a synthesis for the systemic agricultural fungicide tridemorph from dodecanoic acid (lauric acid), propene, and a one-carbon building block. How many stereoisomers are possible for tridemorph? CH3 →CH3(CH2)10COOH + CH3CH=CH, H3C (CH2) 12CH3 Tridemorph Dodecanoic acid Propene (Lauric acid)arrow_forwardA chemist synthesized compound X as a racemic mixture. When the ketone group in X was enzymatically reduced to the corresponding alcohol, a 100% yield was obtained of the product shown below. Choose the statement that best describes this result. ОН enzyme C;H1 `OCH,CH; pH 4.0 C3H1 `OCH,CH3 ОН ÕH X (racemic) (100% yield) One enantiomer of compound X reacts quickly with the enzyme. The other enantiomer of compound X is unreactive, but rapidly equilibrates with the reactive enantiomer under the reaction conditions. Since compound X was racemic, it makes sense that only a single product was obtained. O The product is a meso compound, so either enantiomer of compound X gives the same product. One enantiomer of compound X reacts quickly with the enzyme, while the other enantiomer of compound X remains unchanged.arrow_forward

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

