
Organic Chemistry, 12e Study Guide/Student Solutions Manual
12th Edition
ISBN: 9781119077329
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 13PP
Practice Problem 17.13
(a) Provide the reagents required to accomplish the following transformation.
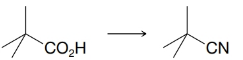
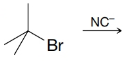
(b) What product would you likely obtain if you attempted to synthesize the nitrile above by the following method?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Suggest reasonable reagents that could be used to carry out the following transformations:
Show how the following ketones might be synthesized from the indicated acids, usingany necessary reagents.(b) methyl cyclohexyl ketone from cyclohexanecarboxylic acid
Give the products expected when acetic formic anhydride reacts with (i) aniline and(ii) benzyl alcohol.(b) Propose mechanisms for these reactions.
Chapter 17 Solutions
Organic Chemistry, 12e Study Guide/Student Solutions Manual
Ch. 17 - Practice Problem 17.1 Give an IUPAC systematic...Ch. 17 - Prob. 2PPCh. 17 - Practice Problem 17.3 Write structural formulas...Ch. 17 - Practice Problem 17.4
Show how each of the...Ch. 17 - Practice Problem 17.5
Show how you could prepare...Ch. 17 - Practice Problem 17.6
(a) Which of the carboxylic...Ch. 17 - Prob. 7PPCh. 17 - Prob. 8PPCh. 17 - Practice Problem 17.9
Esters can also be...Ch. 17 - Prob. 10PP
Ch. 17 - Prob. 11PPCh. 17 - Practice Problem 17.12
What products would you...Ch. 17 - Practice Problem 17.13 (a) Provide the reagents...Ch. 17 - Prob. 14PPCh. 17 - Practice Problem 17.15 Using decarboxylation...Ch. 17 - Practice Problem 17.16 Diacyl peroxides, ,...Ch. 17 - Prob. 17PCh. 17 - Give an IUPAC systematic or common name for each...Ch. 17 - Prob. 19PCh. 17 - Prob. 20PCh. 17 - 17.21 What major organic product would you expect...Ch. 17 - Prob. 22PCh. 17 - Prob. 23PCh. 17 - Prob. 24PCh. 17 - Prob. 25PCh. 17 - 17.26 What products would you expect to obtain...Ch. 17 - Write structural formulas for the major organic...Ch. 17 - 17.28 Indicate reagents that would accomplish each...Ch. 17 - Write structural formulas for the major organic...Ch. 17 - Prob. 30PCh. 17 - Prob. 31PCh. 17 - Prob. 32PCh. 17 - 17.33 On heating,...Ch. 17 - Prob. 34PCh. 17 - Prob. 35PCh. 17 - 17.36 Show how pentanoic acid can be prepared from...Ch. 17 - 17.37 The active ingredient of the insect...Ch. 17 - Prob. 38PCh. 17 - Prob. 39PCh. 17 - Give stereochemical formulas for compounds AQ:...Ch. 17 - 17.41 -Glyceraldehyde can be transformed into...Ch. 17 - Prob. 42PCh. 17 - Prob. 43PCh. 17 - 17.44 Given here are the NMR spectra and carbonyl...Ch. 17 - 17.45 Compound Y dissolves slowly when warmed...Ch. 17 - Prob. 46PCh. 17 - Prob. 47PCh. 17 - Prob. 48PCh. 17 - Prob. 49PCh. 17 - Prob. 50PCh. 17 - Prob. 51PCh. 17 - 17.52 Starting with 1-naphthol, suggest an...Ch. 17 - Suggest a synthesis of ibuprofen (Section 5.11)...Ch. 17 - Prob. 54PCh. 17 - Prob. 55PCh. 17 - Prob. 1LGPCh. 17 - Prob. 2LGPCh. 17 - Prob. 3LGPCh. 17 - Prob. 4LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
12.1 Give the IUPAC name for each of the following:
a. CH3-CH2-OH
b.
c.
d.
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
1. Why is the quantum-mechanical model of the atom important for understanding chemistry?
Chemistry: Structure and Properties (2nd Edition)
For the 3pz and 4dxz hydrogen-like atomic orbitals, sketch the following: a. The radial function R b. The radia...
Inorganic Chemistry
Consider the control volume shown for the special case of steady-state conditions with v=0,T=T(y), and =const. ...
Fundamentals of Heat and Mass Transfer
In this chapter, we have learned about the photoelectric effect and its impact on the formulation of light as ...
Chemistry: The Central Science (14th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Following is a synthesis for toremifene, a nonsteroidal estrogen antagonist whose structure is closely related to that of tamoxifen. (a) This synthesis makes use of two blocking groups, the benzyl (Bn) group and the tetrahydropyranyl (THP) group. Draw a structural formula of each group and describe the experimental conditions under which it is attached and removed. (b) Discuss the chemical logic behind the use of each blocking group in this synthesis. (c) Propose a mechanism for the conversion of D to E. (d) Propose a mechanism for the conversion of F to toremifene. (e) Is toremifene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forwardExplain how benzaldehyde and dimedone reacts with each other, and then with the aminotriazole to form compound 1a in the presence of an acid catalyst. Provide a detailed reaction mechanism and explanation.arrow_forwardShow how you would synthesize the following esters from appropriate acyl chloridesand alcohols. (c) benzyl benzoate (d) cyclopropyl cyclohexanecarboxylatearrow_forward
- In an aqueous solution containing sodium bicarbonate, aniline reacts quickly withbromine to give 2,4,6-tribromoaniline. Nitration of aniline requires very strong conditions,however, and the yields (mostly m-nitroaniline) are poor.(a) What conditions are used for nitration, and what form of aniline is present under theseconditions?arrow_forwardPropose a synthesis for each of the following compounds. (a) OH (b) (c)arrow_forward(a) How will you obtain the following :(i) Benzaldehyde from Phenol (ii) Benzoic acid from Aniline(b) Give reasons :(i) Aldehydes are more reactive than ketones towards nucleophilic reagents.(ii) Electrophilic substitution in benzoic acid takes place at meta position.(iii) Carboxylic acids do not give the characteristic reactions of carbonyl group.arrow_forward
- The following questions pertain to the esters shown and behavior under conditions of the Claisen condensation.(a) Two of these esters are converted to β-keto esters in good yield on treatment with sodium ethoxide and subsequent acidification of the reaction mixture. Which two are these? Write the structure of the Claisen condensation product of each one. (b) One ester is capable of being converted to a β-keto ester on treatment with sodium ethoxide, but the amount of β-keto ester that can be isolated after acidification of the reaction mixture is quite small. Which ester is this? (c) One ester is incapable of reaction under conditions of the Claisen condensation. Which one? Why?arrow_forwardShow how you would synthesize the following esters from appropriate acyl chloridesand alcohols.(a) ethyl propionate (b) phenyl 3-methylhexanoatearrow_forward(a) Explain the mechanism of a nucleophilic attack on the carbonyl group of an aldehyde or a ketone.(b) An organic compound (A) (molecular formula CgH16Q2) was hydrolysed with dilute sulphuric acid to give a carboxylic acid (B) and an alcohol (C). Oxidation of (C) with chromic acid also produced (B). On dehydration (C) gives but-1-ene. Write the equations for the reactions involved.arrow_forward
- Suggest how you would synthesize each compound, use cyclopentanone as one of the reagentsarrow_forwardShow how you would synthesize the following compounds, starting with benzene or toluene and any necessary acyclic reagents. Assume para is the major product (and separable from ortho) in ortho, para mixtures.(a) p-aminobenzoic acid (b) 2-methyl-1-phenylbutan-2-ol (c) 5-chloro-2-methylanilinearrow_forwardPropose mechanisms consistent with the following reactions. (a) (b) (c) (d) (f) (breif Written explantion for each PLEASE) Propose mechanisms consistent with the following reactions. (a) HBr ROOR Br H₂SO4 (b) H₂O OH Br HBr Br (d) (c) CHBr Br NaOH Br Br (e) HCI CH3CH₂OH Br OCH2CH3 Br ED-8 CH2CH3 H- OH H+, H₂O (f) H OH CH3CH2 CH2CH3 CH2CH3 (continued)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY