
Concept explainers
(a)
Interpretation: The structure of given compounds has to be drawn.
ethylacetoacetate
Concept introduction: The
Carboxylate ion with a carbonyl group at the 3-position loses
(a)
Answer to Problem 48P
The structure of ethyl acetoacetae is,
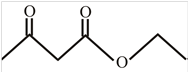
Explanation of Solution
The name given compound ethyl acetoacetate ends with ate. This means the given compound must contain an ester group. The name of compound starts with ethyl group, this means ethyl group is attached to the oxygen of an ester group. The acetone group is attached with the carbonyl carbon of an ester group. The structure of ethyl acetoacetae is,

(b)
Interpretation: The structure of given compounds has to be drawn.
α-methylmalonicacid
Concept introduction: The
Carboxylate ion with a carbonyl group at the 3-position loses
(b)
Answer to Problem 48P
The structure of α-methylmalonic acid is,
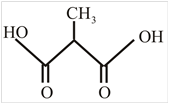
Explanation of Solution
The structure of malonic acid is two

(c)
Interpretation: The structure of given compounds has to be drawn.
β-keto ester
Concept introduction: The
Carboxylate ion with a carbonyl group at the 3-position loses
(c)
Answer to Problem 48P
The structure of β-keto ester is,

Explanation of Solution
The given compound β-keto ester contains a keto group and an ester group. The structure of β-keto ester is,

(d)
Interpretation: The structure of given compounds has to be drawn.
The carboxylic acid obtained from malonic ester synthesis when the
Concept introduction: The
Carboxylate ion with a carbonyl group at the 3-position loses
(d)
Answer to Problem 48P
The structure of enol form of cyclopentanone is,
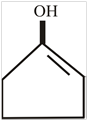
Explanation of Solution
The compound cyclopentanone contain a keto group. After the keto enol tautomerism the keto compound converted into an alcohol and a double bond. The structure of enol form of cyclopentanone is,

(e)
Interpretation: The structure of given compounds has to be drawn.:
The structure of enol form of cyclopentanone
Concept introduction: The
Carboxylate ion with a carbonyl group at the 3-position loses
(e)
Answer to Problem 48P
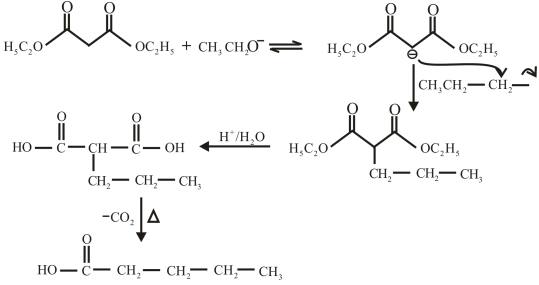
The structure of the carboxylic acid obtained from malonic ester synthesis by using propyl bromide is,
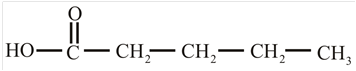
Explanation of Solution
The proton removed from the alpha carbon of malonic ester by the base. Then, there is a nucleophilic substitution reaction takes place between the propyl bromide and carbanion of malonic ester. The third step is acidic hydrolysis of an ester to form carboxylic acid. The fourth step is decarboxylation of the compound to form the desired product. The reaction and structure of given compound is,
Want to see more full solutions like this?
Chapter 17 Solutions
ORGANIC CHEMISTRY-W/S.G+SOLN.MANUAL
- 5. Draw the structure of following compounds: a. methyl butanoate b. propyl ethanoate c. ethyl propanoate d. ethyl 3-methylhexanoate e. butyl hexanoate f. methyl 2-hexyl-3-oxocyclopentanecarboxylatearrow_forwardWhat test will allow you to distinguish between: a. benzyl alcohol and cyclohexanolb. benzyl alcohol and phenol c. cyclohexanol and 1-methylcyclohexanol d. o-cresol and anisolee. benzyl alcohol and anisolearrow_forwardWhich of the following ketone will produce propanoic acid only after oxidation by acidified potassium dichromate? Choices are a. diethyl ketone b. dimethyl ketone c. ethyl n-propyl ketone d. ethyl methyl ketonearrow_forward
- Write the equation for the reaction of the following with acetaldehyde: a. 2,4-DNP b. Schiff's reagant c. Tollen's reagent d. Fehling's solution e. Acidified KMnO4arrow_forwardWhat are the products of the following reactions? (A trace amount of acid is present in each case.) a. cyclopentanone + ethylamine c. acetophenone + hexylamine b. cyclopentanone + diethylamine d. acetophenone + cyclohexylaminearrow_forwardWhich of the following sets of reagents would not be an acceptable method for the preparation of ethylacetate? A. acetic acid, ethanol, and an acid catalyst B. Sodium acetate and ethanol C. Acetic anhydride and ethanol D. Sodium acetate and ethyliodidearrow_forward
- Which of the following products could you not create using malonic acid ethyl ester as a starting material? A. diethyl-2,2-dialkylmalonate B. maleic acid C. Carboxylic acid D. 2,2-dialkylmalonic acid E. diethyl 2-alkyl malonatearrow_forwardWrite the equation of the reaction of the following reagent with acetaldehyde: a. 2,4-DNP b. SCHIFF'S reagent c. Tollen's Reagent d. Fehling's Test e. Acidified KMnO4arrow_forward20. Picric acid does NOT exhibit acidic properties compared to phenol despite the presence of the hydroxyl group because of this structural effect: A. Resonance B. Hyperconjugation C. Electron-attracting Inductive Effect D. Steric effectarrow_forward
- a. What two amides are obtained from the reaction of acetyl chloride with an equivalent of ethylamine and an equivalent of propylamine?b. Why is only one amide obtained from the reaction of acetyl chloride with an equivalent of ethylamine and an equivalent of triethylamine?arrow_forwardWhen phenylbenzoate is heated with aqueous acid solution, the products formed are: A. 2 moles of benzylalcohol B. 1 mole benzoic acid + 1 mole benzylalcohol C. 1 mole benzoic acid + 1 mole phenol D. 2 moles of benzoic acidarrow_forwardNabumetone is a pain reliever and anti-inflammatory agent sold under the brand name of Relafen. a. Write out a synthesis of nabumetone from ethyl acetoacetate. b. What ketone and alkyl halide are needed to synthesize nabumetone by direct enolate alkylation?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
