
Interpretation:
The
Concept introduction:
The carbonyl group
The general reaction of imine fomrtaion is given as,
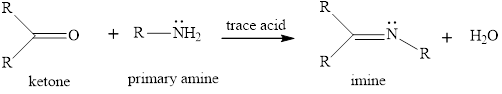
The imine formation is
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
- Using pKa Values to Determine Relative Acidity and Basicity Rank the following compounds in order of increasing acidity, and then rank their conjugate bases in order of increasing basicity.arrow_forwardUse the value of the pK1 provided to deduce the structures of the following oxo acids. Acid pK1 H5IO6 2 H3PO2 2arrow_forwardShow the reaction and equilibrium expression for the basic dissociation of trimethylamine. Given that the Ka = 1.58 x 10-10 for trimethyl ammonium ion, use this to calculate the equilibrium constant for the basic dissociation of trimethyl amine. If the concentration of trimethylamine is 0.10 M, what is the pH of the solution?arrow_forward
- Drug A is a weak acid that has a pKa value of 4. For the Drug A dissolved in Buffer B, calculate what percentage of Drug A is protonated? Can drug A diffuse freely across cell membrane?The pH for Drug A in Buffer B is 6.6 pH. The concentration is 0.1Marrow_forwardDicarboxylic acids have two dissociation constants, one for the initial dissociation into a monoanion and one for the second dissociation into a dianion. For oxalic acid, HO2C—CO2H, the first ionization constant is pKal = 1.2 and the second ionization constant is pKa2 = 4.2. Why is the second carboxyl group far less acidic than the first?arrow_forwardFind the pH of 0.051 M trimethylammomium chloride solution given a pKa of 9.799 for the trimethylammonium ion.arrow_forward
- 1. Write the chemical equation for the acid dissociation of acetaminophen, C8H9O2N. 2. Write the Ka expression for the acid dissociation of acetaminophen.arrow_forward(i) Draw the dissociation reaction for a carboxylic acid in water and define the Ka for this reaction. Write the equation that relates pKa to Ka. (ii) A deprotonated carboxylic acid can be drawn in two resonance forms. Draw the two forms and explain what the term “resonance” means. (iii) Draw an energy profile for the above dissociation reaction and describe how the profiles for a strong and a weak acid would differ.arrow_forwardEstimate the Keq for the acid/base equilibria below using pKa Values. (Not provided so I have been looking in my text book and online and not getting anywhere..)arrow_forward
- 1) Consider the reaction AH(+) + H₂O → A: + H₂O¹. For the following named acids: 1) draw the structure of the acid, 2) give the approximate pKa of the conjugate acid (±1 pKa unit), 3) give the name of the conjugate base, and 4) draw the structure of the conjugate base. For the purposes of answering this question you may use pKa values evenly divisible by 5. Acetylacetone pKa acid Conj. base acid Conj. base iso-Butyl amine pKa Imidazolium cation pKa sec-butyl oxonium pKa t-Butyl Acetoacetate pKa iso-propanol pka_ Ethyl Acetate pKa Anilinium cation pKaarrow_forwardWhat do you mean by carbon acid? What is the range of their pKa's?arrow_forwardThe pKas of the conjugate acids of -OH (conjugate acid H2O) and HPO42- (conjugate acid H2PO4-) are 15.7 and 6.8, respectively. First, identify which is the strong base, and which is the weak base. Then, explain how you can use these pKa values to help you identify which compounds will be separated at each step.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

