
ORGANIC CHEMISTRY MASTERINGCHEM ACCESS
9th Edition
ISBN: 9780137249442
Author: Wade
Publisher: INTER PEAR
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17.14C, Problem 17.32P
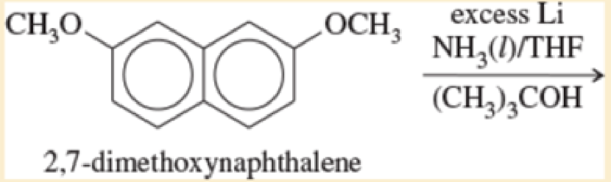
Predict the major products of the following reactions.
- a. toluene+excessCl2 (heat, pressure)
- b. benzamide(PhCONH2)+Na(liquidNH3,CH3CH2OH)
- c. o-xylene+H2 (1000 psi, 100 °C, Rh catalyst)
- d. p-xylene+Na(liquidNH3,CH3CH2OH)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which of the following compounds can be used to synthesize ethanoicpropanoic anhydride
(CH;COOOCCH½CH3)?
A. CH;COCI+ CH;CH2COO`Na* →
B. CH;COOH +heat →
C. CH3CH2CH2COCI + CH;COO Na* →
D. A and C
Predict the Product. Provide the stable organic product(s) for the reactions below.
1. CH,MgBr (xs)
2. HCI, H20
1. NaBH4, H20
2.
MgBr
(1 eq.), THF
1.
MgBr
(1 eq.), THF
2. HCI, НаО
3. O
1. Mg, diethyl ether
Br
2. CH,OD
X
Upon ozonolysis, Compound X produces two compounds: Compound Y and
Compound Z. Compound Y can also be prepared from the following synthetic
route:
PCC
1. R₂BH, THF
1. Mg. Et₂O
PCC
Compound Y
2.
CH₂Cl₂
2. NaOH, HO
CH₂Cl₂
3. H₂O*
From this information, draw the structures of Compounds X, Y, and Z. For
Compounds X and Z, different substituents are possible. For grading purposes,
just use hydrogens as the substituents.
Br
مرد
→]
►
Chapter 17 Solutions
ORGANIC CHEMISTRY MASTERINGCHEM ACCESS
Ch. 17.1 - Prob. 17.1PCh. 17.2 - Prob. 17.2PCh. 17.3 - Prob. 17.3PCh. 17.4 - Use resonance forms to show that the dipolar sigma...Ch. 17.6A - Prob. 17.5PCh. 17.6A - Prob. 17.6PCh. 17.6B - Propose a mechanism for the brommation of...Ch. 17.6B - Prob. 17.8PCh. 17.6B - Prob. 17.9PCh. 17.7 - Prob. 17.10P
Ch. 17.8 - Draw all the resonance forms of the sigma complex...Ch. 17.9 - Predict the mononitration products of the...Ch. 17.9 - Predict the mononitration products of the...Ch. 17.9 - Prob. 17.14PCh. 17.10 - Propose products (if any) and mechanisms for the...Ch. 17.10 - Predict the products (if any) of the following...Ch. 17.10 - Which reactions will produce the desired product...Ch. 17.10 - Prob. 17.19PCh. 17.11C - Prob. 17.20PCh. 17.12A - Prob. 17.21PCh. 17.12B - Propose a mechanism that shows why p-chlorotoluene...Ch. 17.12B - Propose mechanisms and show the expected products...Ch. 17.12B - Prob. 17.24PCh. 17.13A - What products would you expect from the following...Ch. 17.13A - What organocuprate reagent would you use for the...Ch. 17.13B - What products would you expect from the following...Ch. 17.13B - Prob. 17.28PCh. 17.13C - What products would you expect from the following...Ch. 17.13C - Prob. 17.30PCh. 17.14C - Prob. 17.31PCh. 17.14C - Predict the major products of the following...Ch. 17.15A - Predict the major products of treating the...Ch. 17.15B - Prob. 17.34PCh. 17.15B - Prob. 17.35PCh. 17.15B - Predict the major products when the following...Ch. 17.15C - Prob. 17.37PCh. 17.15C - a. Based on what you know about the relative...Ch. 17.15C - Show how you would synthesize the following...Ch. 17.16A - The bombardier beetle defends itself by spraying a...Ch. 17.16B - Predict the products formed when m-cresol...Ch. 17.16B - Prob. 17.42PCh. 17.16B - Prob. 17.43PCh. 17.16B - Predict the site(s) of electophilic attack on...Ch. 17.16B - Prob. 17.45PCh. 17.16B - Prob. 17.46PCh. 17.16B - Propose a synthetic sequence of this...Ch. 17.16B - Prob. 17.48PCh. 17.16B - Starting from toluene, propose a synthesis of this...Ch. 17 - Prob. 17.50SPCh. 17 - Prob. 17.51SPCh. 17 - Show how you would synthesize the following...Ch. 17 - Predict the major products of the following...Ch. 17 - Predict the major products of bromination of the...Ch. 17 - What products would you expect from the following...Ch. 17 - Prob. 17.56SPCh. 17 - Prob. 17.57SPCh. 17 - The following compound reacts with a hot,...Ch. 17 - Prob. 17.59SPCh. 17 - Electrophilic aromatic substitution usually occurs...Ch. 17 - Prob. 17.62SPCh. 17 - The most common selective herbicide for killing...Ch. 17 - Furan undergoes electrophilic aromatic...Ch. 17 - Prob. 17.65SPCh. 17 - Bisphenol A is an important component of many...Ch. 17 - Prob. 17.67SPCh. 17 - Prob. 17.68SPCh. 17 - Prob. 17.69SPCh. 17 - In Chapter14, we saw that Agent Orange contains...Ch. 17 - Phenol reacts with three equivalents of bromine in...Ch. 17 - Prob. 17.72SPCh. 17 - Prob. 17.73SPCh. 17 - A common illicit synthesis of methamphetamine...Ch. 17 - Prob. 17.75SPCh. 17 - Prob. 17.76SPCh. 17 - Prob. 17.77SPCh. 17 - Prob. 17.78SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 1. Provide IUPAC names of the following compounds. a. CHỊCH,CH,CHO .or C. CHO محمد 2. predict the major products of the following reactions. 8 CN COOH PHNHNH, H* 1) DIBAL-H 2) H₂O* 1. SOCI₂ 2. [(CH₂)₂CHCH₂₂Culi 3. H₂Oarrow_forwardWhich structural features can undergo pinacol-pinacolone type of reactions? CH3CH(OH)CH(NH2)CH3 ОНОН OH II II A. I and II B. Il and III C. I and III D. I, II, and IIlarrow_forwardGive the organic reaction products for the following reaction a. b. a. b. CH3CH2-C. ง ว NaOH ? + ? N(CH3)2 H₂O H*/H₂O CH3-C-OCH2CH3 ? + ?arrow_forward
- 1. Write the structure of the major organic product for reactions a, b and e? H₂C b. H₂c- CH, CH₂ Cold KMnO, OH A KMnO, H c. CH3CH₂-CH=CH-CH₂CH3 + H₂SO4 →arrow_forwardComplete the roadmap below by providing the correct reagents for each conversion from the list. B. C. D. G H. J. H 1. PCC in pyridine 2. ethanol with catalytic sulfuric acid 3. NBS, hv 4. LDA with propanal 1. PPh3, DMF 5. 2. nbuli 3. 2-propanone 6. NaOH 7. H₂Cr₂07 8. methylpropanoate, NaOCH3 in methanol 9. CH₂CH₂MgBr then aqueous H₂SO4 10. mCPBAarrow_forwardChemistry 3. The following sequence generates crystals. The crystals produced are insoluble in organic solvents but freely soluble in water. They have a melting point of 203 °C. There is an instantaneous precipitate formed with silver nitrate solution. Catalytic hydrogenation gives cycloheptane. What is the crystalline compound? Why does it have these unexpected properties? heat 1 equiv Br2 dibromide crystals - HBr tropilidenearrow_forward
- Choose the two alkyl bromides and the hydrogenation method that must be used to synthesize these alkenes from acetylene. Enter your answer as two letters in alphabetical order, followed by a number; i.e. ac2, not ca2. Do not use punctuation. Alkyl Halides a. CH3CH,Br. b. CH3(CH2)4Br c. CH3(CH2);Br d. CH3(CH2)6Br e. CH3(CH2);Br f. CH3(CH2);Br g. CH3(CH2)12B Hydrogenation Method 1. H2, Lindlar's catalyst 2. Na, NH3 (1) Alkene #1: H3C CH3 Alkene #2: CH3 H3Carrow_forwardProvide the structural formula of the products that are formed when acetophenone reacts with these reagents: 1. NaBH4 in Methanol 2. Ag(NH3)2+ 3. C6H5MgBr, then H3O+ 4. H2 in Pt 5. Hydroxylamine 6. Phenylhydrazine 7. LiAlH4 then water 8. HCN 9. Diethylamine 10. 2 CH3OH, HCl catalystarrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b. CH3COCl c. (CH3CO)2O d. excess CH3I e. (CH3)2C = O f. CH3COCl, AlCl3 g. CH3CO2H h. NaNO2, HCl i. Part (b), then CH3COCl, AlCl j. CH3CHO, NaBH3CNarrow_forward
- Claisen condensation between diethyl phthalate and ethyl acetate followed by saponifi- cation, acidification, and decarboxylation forms a diketone, C,H,O,. COOEt 1. EtO Na+ HCI, H,O B NaOH, H,O + CH3COOEt C,H,O2 2. НС, Н,О heat heat COOEt Diethyl phthalate Ethyl acetate Propose structural formulas for compounds A and B and the diketone.arrow_forward1. write out all the isomers of the compound with molecular formula C4H10O. 2. select the normal/primary isomer and treat it with conc.H2SO4 and heat to produce A. Identify the type of reaction 3. Treatment of A with HCl/H2O gives B and with cold KMnO4/ OH- gives C . Give the name and structures of A, B and C 4. Treatment of A with Hot KMnO4/OH- gives D followed by acidification of the mixture to give E. What is D and E ?arrow_forwardBy means of a series of equations outline a method for carrying out the ff. conversions. You may use reagents you wish. More than one step maybe required. a. acetylene b. cyclohexanone c. ethyl bromide d. benzene athanol +methylcyclohexanol acetaldehyde +phenyl ethanol B-ethyl-3-pentanol tyclopentanone 2-суano-2-pentanol benzyl alcohol 2-butanone-semicarbazone е. 3-рentanol f. cyclopentene g. 2-pentanol h. benzene i. 2-butanolarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY