
Interpretation:
A synthesis of diethylcarbamazine from the given starting materials has to be given.
Concept introduction:
Retro synthesis: It is a technique of planning an
Base catalysed ring opening of
The nucleophile will attack at the less substituted position under basic conditions and then the alkoxide ion gets proton from alcohol which form the product.
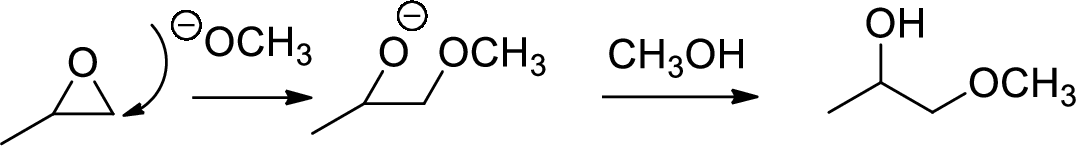
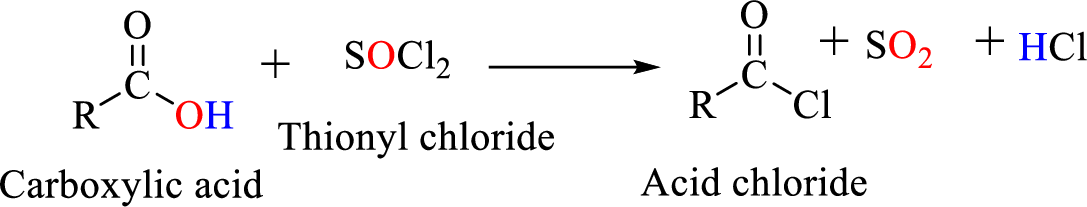
Acid chlorides are most often prepared by treating a
Chloroformates:
They are the class of organic compounds which are colourless and having a formula
Reaction of Chloroformates with
The product will be carbamates.
Trending nowThis is a popular solution!

Chapter 18 Solutions
ORGANIC CHEMISTRY-OWL V2 ACCESS
- Phenacetin is an analgesic compound having molecular formula C10H13NO2. Once a common component in over-the-counter pain relievers such as APC (aspirin, phenacetin, caffeine), phenacetin is no longer used because of its liver toxicity. Deduce the structure of phenacetin from its 1H NMR and IR spectra.arrow_forwardThe two most general amine syntheses are the reductive amination of carbonyl compounds and the reduction of amides.Show how these techniques can be used to accomplish the following syntheses.(a) benzoic acid S benzylamine (b) benzaldehyde S benzylamine(c) pyrrolidine S N@ethylpyrrolidine (d) cyclohexanone S N@cyclohexylpyrrolidine(e) HOOC¬(CH2)3 ¬COOH S pentane@1,5@diamine (cadaverine)arrow_forwardGiven this retrosynthetic analysis, propose a synthesis for bupropion.arrow_forward
- Reaction of p-nitroaniline with sodium nitrite and hydrochloric acid at 0°C, followed by treatment with N,N-diethylaniline.arrow_forwardFollowing is a synthesis for toremifene, a nonsteroidal estrogen antagonist whose structure is closely related to that of tamoxifen. (a) This synthesis makes use of two blocking groups, the benzyl (Bn) group and the tetrahydropyranyl (THP) group. Draw a structural formula of each group and describe the experimental conditions under which it is attached and removed. (b) Discuss the chemical logic behind the use of each blocking group in this synthesis. (c) Propose a mechanism for the conversion of D to E. (d) Propose a mechanism for the conversion of F to toremifene. (e) Is toremifene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forwardCompound H (C8H6O3) gives a precipitate when treated with hydroxylamine in aqueous ethanol and a silver mirror when treated with Tollens solution. Following is its 1H-NMR spectrum. Deduce the structure of compound H.arrow_forward
- Barbiturates are prepared by treatment of diethyl malonate or a derivative of diethyl malonate with urea in the presence of sodium ethoxide as a catalyst. Following is an equation for the preparation of barbital, a long-duration hypnotic and sedative, from diethyl diethylmalonate an urea. Barbital is prescribed under one of a dozen or more trade names. Propose a mechanism for This reaction The pKa of barbital is 7.4. Which is the most acidic hydrogen in this molecule and how do you account for its acidity?arrow_forwardThe following questions concern ethyl (2-oxocyclohexane)carboxylate.(a) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by a Dieckmann cyclization.(b) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by acylation of a ketone.(c) Write structural formulas for the two most stable enol forms of ethyl (2-oxocyclohexane)carboxylate.(d) Write the three most stable resonance contributors to the most stable enolate derived from ethyl (2-oxocyclohexane)carboxylate.(e) Show how you could use ethyl (2-oxocyclohexane)carboxylate to prepare 2-methylcyclohexanone.(f) Give the structure of the product formed on treatment of ethyl (2-oxocyclohexane)-carboxylate with acrolein (H2C=CHCH=O) in ethanol in the presence of sodium ethoxidearrow_forwardReaction of p-nitroaniline with sodium nitrite and hydrochloric acid at 0 °C, followed by treatment with N,N-diethylaniline.arrow_forward
- Atenolol is a beta blocker medication primarily used to treat high blood pressure and heart- associated chest pain. Other uses include the prevention of migraines and treatment of certain irregular heart beats It is taken by mouth or by injection into a vein. It can also be used with other blood pressure medications. OH .N. CH3 ČH3 H2N° suggest industrial synthetic approach towards synthesis of this drug (atenolol) and provide reasons and advantages of your proposed synthetic approach.arrow_forwardNonearrow_forwardDiscovery of the antibiotic sulphanilamide led to rapid development of a large number of structurally related sulphonamides. Some of these were useful leads to compounds with other medicinal properties. Amongst these, sulfasalazine was active in the treatment of ulcerative colitis, a potentially fatal disease of the colon. As a medicinal chemist, you are about to carry out the synthesis of sulfasalazine, starting from aniline. Give the chemical names and draw the chemical structures of the reagents you will need to use for the all the steps marked (a)-(d). Aniline HO₂C HO (a) N=N- ACHN Ac represents CH3CO Sulfasalazine SO,NH S0,C1 N (b) (d) H₂N- SO,NH (c) N [Oro] SO,NH N Step (a) in question 5 above results in the para product only. Explain why this occurs.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


