
Concept explainers
Interpretation: The structures of cyclohexa-2, 4-dien-1-one and its enol form are to be written, and the special factors that account for the stability of the enol form are to be identified.
Concept introduction:
Tautomerization is defined as the interconversion of the keto and enol forms. The keto and enol forms are tautomers.
The conjugate base of both the keto and the enol forms is enolate.
If the enolate accepts a proton from carbon, the keto form is formed, and if the enolate accepts a proton from oxygen, the enol form is formed.
Answer to Problem 1PP
Solution:
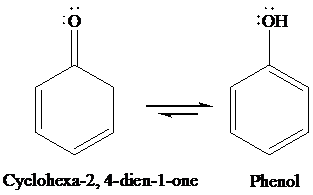
Due to aromaticity.
Explanation of Solution
Under acidic condition, cyclohexa-2, 4-dien-1-one converts to phenol through tautomerism. The structures of cyclohexa-2, 4-dien-1-one and its enol form are as follows:
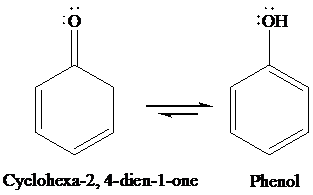
Want to see more full solutions like this?
Chapter 18 Solutions
ORGANIC CHEM.I-W/STD.GDE.+...>CUSTOM<
- When the nitrogen-containing aromatic heterocyclic compounds 1 and 2 are treated with HCl, only 1 forms the hydrochloride salt, whereas compound 2 is unreactive. Provide an explanation for this observed reactivity.arrow_forwardExplain the following observation. Ethyl 3-phenylpropanoate (C6H5CH2CH2CO2CH2CH3) reacts with electrophiles to afford ortho- and para-disubstituted arenes, but ethyl 3-phenylprop-2-enoate (C6H5CH= CHCO2CH2CH3) reacts with electrophiles to afford meta-disubstituted arenes.arrow_forwardExplain why pyridine N-oxide G can undergo nucleophilic aromatic substitution with nucleophile Nuc– to give product I. Include the resonance structures of pyridine N-oxide G to illustrate your answer.arrow_forward
- Compound A produce compound D while undergo Friedel Crafts Alkylation. Compound D is then oxidized and produce compound E (C11H12O3) as a major product.What are the possible structural formula of compound D and E?arrow_forwardStarting with benzene, toluene, or phenol as the only sources of aromatic rings, show how to synthesize the following. Assume in all syntheses that mixtures of ortho-para products can be separated into the desired isomer. Q.) m-Chlorobenzoic acidarrow_forward(−)-Hyoscyamine, an optically active drug used to treat gastrointestinal disorders, is isolated from Atropa belladonna, the deadly nightshade plant, by a basic aqueous extraction procedure. If too much base is used during isolation, optically inactive material is isolated. (a) Explain this result by drawing a stepwise mechanism. (b) Explain why littorine, an isomerisolated from the tailower plant in Australia, can be obtained optically pure regardless of the amount of base used during isolation.arrow_forward
- Wolff-Kishner reduction of compound W gave compound A. Treatment of A with m-chloroperbenzoic acid gave B which on reduction with LiAH4 gave C. Oxidation of compound C with chromic acid gave D (C9H14O). Suggest the structures for A, B, C, and D.arrow_forwardBoth pyridine and pyrrole are nitrogen containing aromatic heterocyclic compounds. When treated with HCl, only pyridine forms the hydrochloride salt, where as pyrrole is unreactive. What is the best explanation for this observed reactivity.arrow_forwardThe endiandric acids comprise a group of unsaturated carboxylic acidsisolated from a tree that grows in the rainforests of eastern Australia.The methyl esters of endiandric acids D and E have been prepared frompolyene Y by a series of two successive electrocyclic reactions: thermalring closure of the conjugated tetraene followed by ring closure of theresulting conjugated triene. (a) Draw the structures (includingstereochemistry) of the methyl esters of endiandric acids D and E. (b)The methyl ester of endiandric acid E undergoes an intramolecular [4 +2] cycloaddition to form the methyl ester of endiandric acid A. Propose apossible structure for endiandric acid A.arrow_forward
- The compound below is treated with chlorine in the presence of lightarrow_forwardGive mechanistic explanations for of the following synthetic transformations. You should explain any regio- or stereochemical control observed as appropriate.arrow_forwardA certain compound is known to contain an aromatic benzene ring but failed to produce a fragrant yellow solution upon subjecting it to the nitration test. What may be a possible explanation for this? A. The benzene ring is part of a highly conjugated, blue dye molecule. B. The benzene ring contains a strong electron-withdrawing group. C. The benzene ring has no available sites left for electrophilic attack. D. All of the given. Kindly explain your answer in detail.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
