
(a) Write structural formulas and provide IUPAC names for all the isomeric
(b) Which of the isomers in part (a) yield chiral alcohols on reaction with sodium borohydride?
(c) Which of the isomers in part (a) yield chiral alcohols on reaction with methylmagnesium iodide?
Interpretation:
The given parts are to be explained.
Concept Introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of the organic compound is correctly interpreted from its name.
Rules for writing the structural formula from IUPAC are as follows:
First, identify the word root for the given compound.
The suffix used in the compound like
The position and location of the substituents that are bonded to the carbon chain must
identified and number theses substituents on the basis of their priorities.
Answer to Problem 23P
Solution:
a)
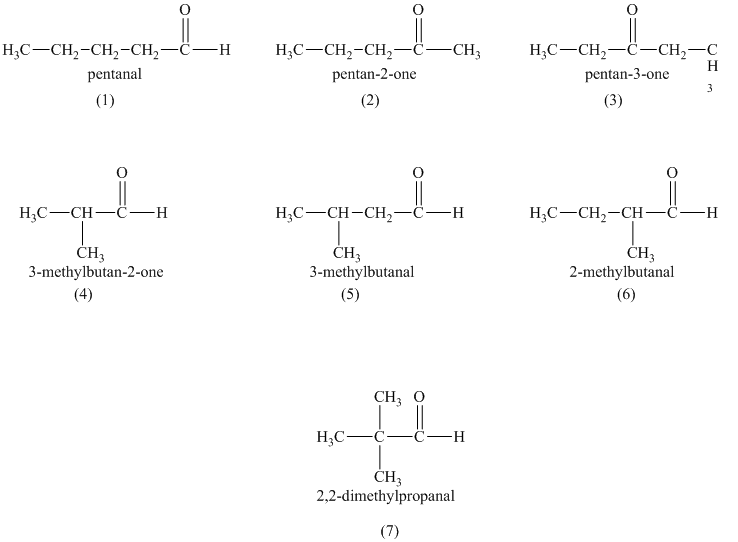
b)
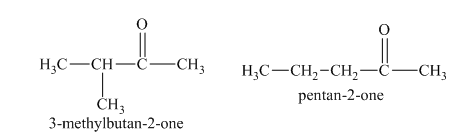
c)
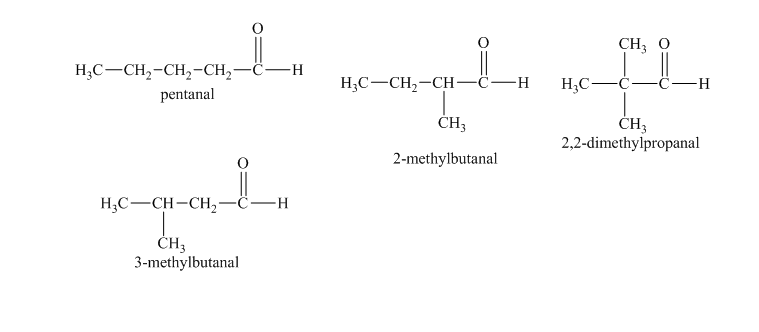
Explanation of Solution
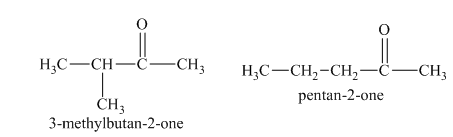
a) Structural formulas and IUPAC name of the isomeric aldehydes and ketones of
The constitutional isomers are the isomers that consist of the same chemical formula but the way they attach to their atoms are totally different from each other.
Aldehydes and ketones contain carbonyl
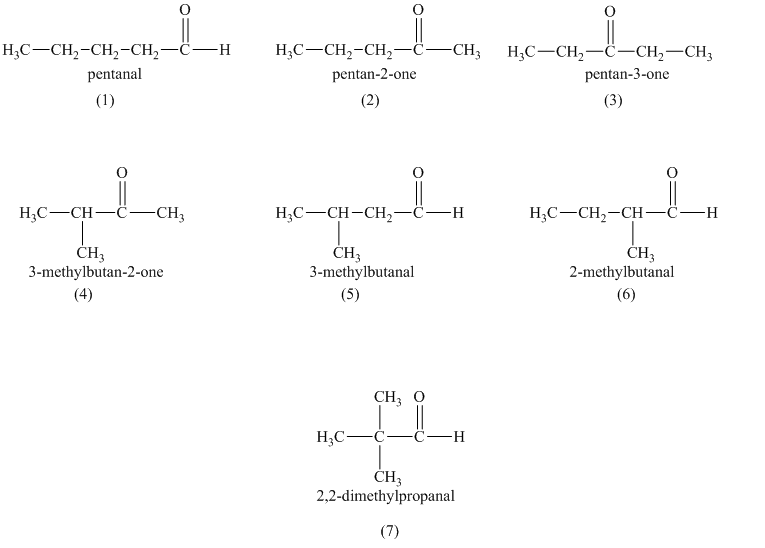
b) The isomers that form chiral alcohols on the reaction with sodium borohydride.
The reduction of aldehydes with sodium borohydride results in the formation of a primary alcohol, whereas in ketones the formation of secondary alcohols is taken place. Primary alcohols are not chiral in nature. On the other hand, the secondary alcohols that are formed from unsymmetrical ketones show chirality. Therefore, among the isomers in part (a),
c) The isomers that give chiral alcohols on reaction with methylmagnesium iodide.
Methylmagnesium iodide is also known as the Grignard reagent. Aldehydes can give chiral alcohols on reaction with methylmagnesium iodide. The formation of chiral and achiral molecules from the isomers that are present in part(a) are shown as:
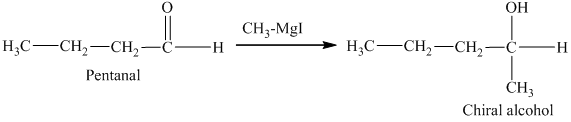
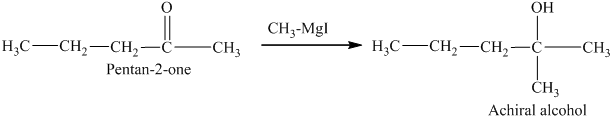
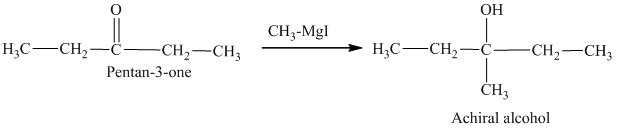
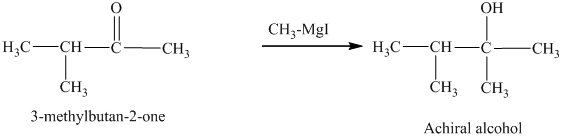
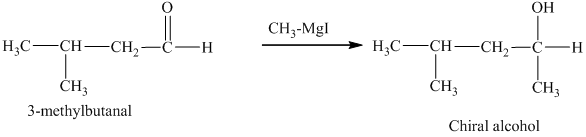
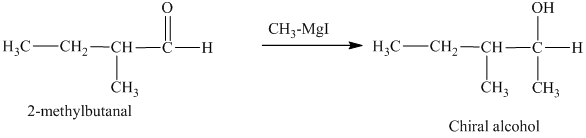
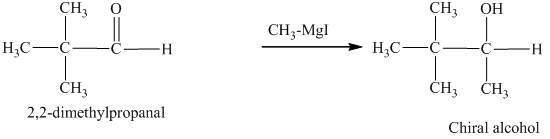
Hence, the isomers that form chiral alcohols on reaction with methylmagnesium iodide are shown below.

Want to see more full solutions like this?
Chapter 18 Solutions
ORGANIC CHEMISTRY(LL)-W/ACCESS >CUSTOM<
- Write structural formulas for all aldehydes with the molecular formula C6H12O and give each its IUPAC name. Which of these aldehydes are chiralarrow_forwardCompound A and compound B are different alcohols with the same molecular formula of C4H10O. When reacted with chromic acid (H2CrO4), compound A and compound B produce compound C and compound D, respectively. Compound C has a molecular formula of C4H8O2. Compound E is obtained when compound A reacts with pyridinium chlorochromate (PCC) in a solvent such as dichloromethane (CH2Cl2). Draw the structure of compound A, B, C, D and E.arrow_forwardDescribe each highlighted bond in terms of the overlap of atomic orbitals. and Draw the structures of ALL of the aldehydes with the molecular formula C5H10O that contain a 5-carbon chain.arrow_forward
- Diazomethane, CH2N2, is used in the organic chemistry laboratory despite its danger because it produces very high yields and is selective for reaction with carboxylic acids. Write the products of the following reactions.arrow_forwardWhat alkane, with molecular formula C7H16, forms seven monochlorinated products (disregarding stereoisomers) when heated with Cl2?arrow_forwardAn unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a palladium catalyst. Hydrocarbon A also reacts with OsO4 to give diol B. When oxidized with KMnO4 in acidic solution, A gives two fragments. One fragment is propanoic acid, CH3CH2CO2H, and the other fragment is ketone C. What are the structures of A, B, and C? Write all reactions, and show your reasoning.arrow_forward
- Compound A, C5H11Br, on treatment with alcoholic KOH, gives two isomeric compoundsarrow_forwardPlease help in answering the following questions ~Which of the following is the basis of the classification of alcohols?A. Based on the number of hydroxyl groups attached.B. Based on the number of carbon atoms which are directly attached to the carbon thatis bonded with the -OH group.C. BothD. Neither ~Which of the following carboxylic acids will yield the least amount of protons in an aqueoussolution?A. 2-chlorobutanoic acidB. Propanoic acidC. Butanoic acidD. 2-methylbutanoic acidarrow_forwardDescribe the procedures on how will you prepare an alcohol from an alkene.arrow_forward
- Draw the structures for and give the names for each of the isomeric carbonyl compounds corresponding to C4H8O and suggest suitable reactions for distinguishing between themarrow_forwardUsing condensed structural formulas, write a balancedchemical equation for each of the following reactions:(a) hydrogenation of cyclohexene, (b) addition of H2O totrans-2-pentene using H2SO4 as a catalyst (two products),(c) reaction of 2-chloropropane with benzene in the presenceof AlCl3.arrow_forwardWhich alkene would the following alcohols form upon dehydration reaction using POCl3 inpyridine? Indicate the major productarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





