
EBK PHYSICS
5th Edition
ISBN: 8220103026918
Author: Walker
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 18.6, Problem 6EYU
Enhance Your Understanding
(Answers given at the end of the chapter)
6.
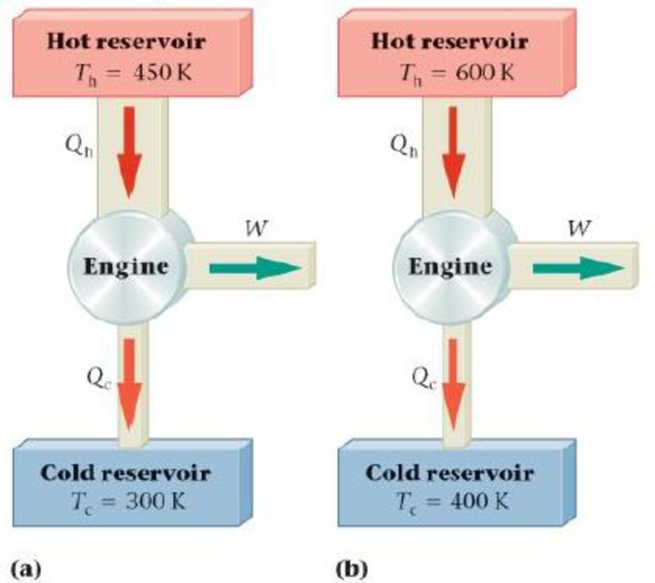
Figure 18-19
Enhance Your Understanding 6.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
True and False: If the statement is true, write "true." If it is false, change the underlined word or
words to make the statement true. Write your answer on the line provided.
1. Real gases behave like ideal gases except at very high temperatures.
2. The gas constant, R, is equal to 0.0821 when the pressure is expressed in
kilopascals.
3. As more gas particles are added to a container, there are fewer collisions
because the particles don't go as far.
4. The number of moles of a gas is inversely proportional to its volume at
STP.
5. Real gases behave like ideal gases except at very high pressure.
6. At a constant temperature, the pressure exerted by one mole of a gas
decreases if the volume available is increased.
7. The ideal gas equation will only give correct values if the temperature in
expressed in degrees Celcius.
8. One mole of oxygen at 760. mm Hg and 0 °C occupies a volume of one L.
Matching: Match each variable listed on the left with the appropriate unit of measurement listed…
Would like answers for part B and C ONLY please-need info from part a to answer them. I already solved part a)
1)Energy Efficiency at Home : Suppose you have two kettles – a plug-in electrickettle and a stovetop kettle. The electric kettle uses electricity from a natural gas fired powerplant, while you boil water in your stovetop kettle on a natural gas burner.
a)Based on the following information, which of these kettles demonstrates a higher energy efficiency (i.e., which is more efficient at using energy from fuel to heat water)? Please calculate the efficiencies of each kettle and express answers as percentages.
-The natural gas power plant converts chemical energy of the natural gas to electrical energy with 58% efficiency.
-High-voltage power lines from the power plant to your house convey electricity with 92% efficiency.
-The electric kettle converts electrical energy to thermal energy in the water with 85% efficiency (the other 15% heats up the kettle itself).
-The stove…
Answer the following questions. Show your complete solution if necessary.
1. Jogging every day is good for your health. Assume that when you jog a work of 500 kJ is done and 230 kJ of heat is given off. What is the change in the internal energy of your body?
2. What is the change in the internal energy of a system when a total of 150.00 J is transferred by heat from the system and 159.00 J is done by work on the system?
Chapter 18 Solutions
EBK PHYSICS
Ch. 18.1 - System 1 is at 0 C and system 2 is at 0 F. If...Ch. 18.2 - Enhance Your Understanding (Answers given at the...Ch. 18.3 - Enhance Your Understanding (Answers given at the...Ch. 18.4 - Enhance Your Understanding (Answers given at the...Ch. 18.5 - Enhance Your Understanding (Answers given at the...Ch. 18.6 - Enhance Your Understanding (Answers given at the...Ch. 18.7 - Enhance Your Understanding (Answers given at the...Ch. 18.8 - Enhance Your Understanding (Answers given at the...Ch. 18.9 - Enhance Your Understanding (Answers given at the...Ch. 18.10 - Enhance Your Understanding (Answer given at the...
Ch. 18 - Prob. 1CQCh. 18 - Heat is added to a substance. Is it safe to...Ch. 18 - Are there thermodynamic processes in which all the...Ch. 18 - An ideal gas is held in an insulated container at...Ch. 18 - Prob. 5CQCh. 18 - Which law of thermodynamics would be violated if...Ch. 18 - Heat engines always give off a certain amount of...Ch. 18 - Prob. 8CQCh. 18 - Which law of thermodynamics is most pertinent to...Ch. 18 - Which has more entropy: (a) popcorn kernels, or...Ch. 18 - Prob. 1PCECh. 18 - A gas expands, doing 100 J of work. How much heat...Ch. 18 - A swimmer does 7.7 105 J of work and gives off...Ch. 18 - When 1310 J of heat are added to one mole of an...Ch. 18 - Three different processes act on a system. (a) In...Ch. 18 - A container holds a gas consisting of 2.85 moles...Ch. 18 - The Charge on Adhesive Tape When adhesive tape is...Ch. 18 - Predict/Calculate One mole of an ideal monatomic...Ch. 18 - Prob. 9PCECh. 18 - A cylinder contains 4.0 moles of a monatomic gas...Ch. 18 - An ideal gas is taken through the three processes...Ch. 18 - Figure 18-26 shows three different multistep...Ch. 18 - Prob. 13PCECh. 18 - An ideal gas is compressed at constant pressure to...Ch. 18 - As an ideal gas expands at constant pressure from...Ch. 18 - A system consisting of an ideal gas at the...Ch. 18 - Prob. 17PCECh. 18 - (a) Find the work done by a monatomic ideal gas as...Ch. 18 - Prob. 19PCECh. 18 - Predict/Calculate If 9.50 moles of a monatomic...Ch. 18 - Suppose 118 moles of a monatomic ideal gas undergo...Ch. 18 - A weather balloon contains an ideal gas and has a...Ch. 18 - Prob. 23PCECh. 18 - During an adiabatic process, the temperature of...Ch. 18 - An ideal gas follows the three-part process shown...Ch. 18 - With the pressure held constant at 260 kPa, 43 mol...Ch. 18 - Prob. 27PCECh. 18 - A system expands by 0.75 m3 at a constant pressure...Ch. 18 - Prob. 29PCECh. 18 - A certain amount of a monatomic ideal gas...Ch. 18 - An ideal gas doubles its volume in one of three...Ch. 18 - Predict/Explain You plan to add a certain amount...Ch. 18 - Find the amount of heat needed to increase the...Ch. 18 - (a) If 585 J of heat are added to 49 moles of a...Ch. 18 - A system consists of 3.5 mol of an ideal monatomic...Ch. 18 - Find the change in temperature if 170 J of heat...Ch. 18 - Gasoline Ignition Consider a short time span just...Ch. 18 - Prob. 38PCECh. 18 - Prob. 39PCECh. 18 - A monatomic ideal gas is held in a thermally...Ch. 18 - Consider the expansion of 60.0 moles of a...Ch. 18 - A Carnot engine can be operated with one of the...Ch. 18 - What is the efficiency of an engine that exhausts...Ch. 18 - An engine receives 660 J of heat from a hot...Ch. 18 - A Carnot engine operates between the temperatures...Ch. 18 - A nuclear power plant has a reactor that produces...Ch. 18 - At a coal-burning power plant a steam turbine is...Ch. 18 - Predict/Calculate A portable generator produces...Ch. 18 - Predict/Calculate The efficiency of a particular...Ch. 18 - During each cycle a reversible engine absorbs 3100...Ch. 18 - Prob. 51PCECh. 18 - The operating temperatures for a Carnot engine are...Ch. 18 - A certain Carnot engine takes in the heat Qh and...Ch. 18 - Predict/Explain (a) If the temperature in the...Ch. 18 - The refrigerator in your kitchen does 490 J of...Ch. 18 - A refrigerator with a coefficient of performance...Ch. 18 - Prob. 57PCECh. 18 - Prob. 58PCECh. 18 - An air conditioner is used to keep the interior of...Ch. 18 - A reversible refrigerator has a coefficient of...Ch. 18 - A freezer has a coefficient of performance equal...Ch. 18 - Predict/Explain (a) If you rub your hands...Ch. 18 - Predict/Explain (a) An ideal gas is expanded...Ch. 18 - Predict/Explain (a) A gas is expanded reversibly...Ch. 18 - Find the change in entropy when 1.85 kg of water...Ch. 18 - Determine the change in entropy that occurs when...Ch. 18 - Prob. 67PCECh. 18 - On a cold winters day heat leaks slowly out of a...Ch. 18 - An 88-kg parachutist descends through a vertical...Ch. 18 - Predict/Calculate Consider the air-conditioning...Ch. 18 - A heat engine operates between a high-temperature...Ch. 18 - It can be shown that as a mass m with specific...Ch. 18 - Prob. 73GPCh. 18 - Figure 18-34 Problem 74 74 CE An ideal gas has...Ch. 18 - The heat that goes into a particular Carnot engine...Ch. 18 - Predict/Calculate Consider 132 moles of a...Ch. 18 - Prob. 77GPCh. 18 - Prob. 78GPCh. 18 - Predict/Calculate Engine A has an efficiency of...Ch. 18 - Nuclear Versus Natural Gas Energy Because of...Ch. 18 - A freezer with a coefficient of performance of...Ch. 18 - Entropy and the Sun The surface of the Sun has a...Ch. 18 - Prob. 83GPCh. 18 - A cylinder with a movable piston holds 2.95 mol of...Ch. 18 - Making Ice You place 0.410 kg of cold water inside...Ch. 18 - An inventor claims a new cyclic engine that uses...Ch. 18 - Predict/Calculate A small dish containing 530 g of...Ch. 18 - Predict/Calculate An ideal gas is taken through...Ch. 18 - One mole of an ideal monatomic gas follows the...Ch. 18 - When a heat Q is added to a monatomic ideal gas at...Ch. 18 - The Carnot Cycle Figure 18-36 shows an example of...Ch. 18 - A Carnot engine and a Carnot refrigerator operate...Ch. 18 - Prob. 93PPCh. 18 - Energy from the Ocean Whenever two objects are at...Ch. 18 - Prob. 95PPCh. 18 - Energy from me Ocean Whenever two objects are at...Ch. 18 - Predict/Calculate Referring to Example 18-21...Ch. 18 - Predict/Calculate Referring to Example 18-21...
Additional Science Textbook Solutions
Find more solutions based on key concepts
7. A factory worker moves a 30.0 kg crate a distance of 4.5 m along a level floor at constant velocity by pushi...
College Physics (10th Edition)
Choose the best answer to each of the following. Explain your reasoning. The distance to the cosmological horiz...
Cosmic Perspective Fundamentals
The correct option.
Glencoe Physical Science 2012 Student Edition (Glencoe Science) (McGraw-Hill Education)
Laptop computers are equipped with accelerometers that sense when the device is dropped and then put the hard d...
Essential University Physics: Volume 1 (3rd Edition)
The enthalpy of combustion of a gallon (3.8 liters) of gasoline is about 31,000 kcal. The enthalpy of combustio...
An Introduction to Thermal Physics
Fill in each blank with the most reasonable metric unit (km, m, cm, or mm).
22. The length of a football field ...
Applied Physics (11th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- A person consumes about 2200 Cal a day. 1 Cal = 4186 J, and 1 kWh = 860 Cal. Part A What is this energy in joules? Express your answer using two significant figures. for Part A for Part A do for Part redo for Part A reset for Part A keyboard shortcuts for Part A help for Pal 2=9.6.106 Submit Previous Answers Request Answer X Incorrect; Try Again; One attempt remaining Review your calculations and make sure you round to 2 significant figures in the last step.arrow_forwardProblem 6 handwriting solve on one page dont do its in half half steps images ok.An inventor claims to have developed a device that executes a power cycle while operating between reservoirs at 900 and 300 K that has a thermal efficiency of (a) 70%, (b) 50%. Evaluate the claim for each case...arrow_forwardAnswer the following questions and be sure to show all your work. a. It is difficult to extinguish a fire on a crude oil tanker, because each liter of crude oilreleases 2.80×10^7 J of energy when burned. To illustrate this difficulty, calculate thenumber of liters of water that must be expended to absorb the energy released byburning 1.00 L of crude oil, if the water has its temperature raised from 20.0ºC to 100ºC,it boils, and the resulting steam is raised to 300ºC.b. Discuss additional complications caused by the fact that crude oil has a smaller densitythan water.arrow_forward
- Problem 2 Calculate the specific heat of Lead (Pb), using both the Einstein model and the Debye model, for temperatures equal to 2, 10, 20, 50, 100, 200, 300, 600, and 800 K. Use ] = 88 K and = 65 K since the specific heat calculated with these values agree with the data well for the whole temperature range. Compare your answer with the values from Touloukian and Buyco*. Explain the low-temperature and high-temperature behavior. *Y.S. Touloukain and E.H. Buyco, Thermophysical Properties of Matter, Vol. 4: Specific Heat-Metallic Elements and Alloys; Vol. 5: Specific Heat-Nonmetallic Solids, IFI/Plenum, New York, 1970.arrow_forwardWrite your solution and answer correctly. 1. A 5 kg ball was thrown at the height of 10 meters with a speed of 18 m/s from rest. Calculate the amount of energy present in the ball.arrow_forwardQuestion 2 A particular power plant operates with a heat-source reservoir at 350°C and a heatsink reservoir at 30°C. It has a thermal efficiency equal to 55% of the Carnot-engine thermal efficiency for the same temperatures. What is the thermal efficiency of the plant? Use the editor to format your answerarrow_forward
- In CHRONOLOGICAL order, the names of the prominent scientists, when, and what they contributed to the development of Thermodynamics. Please cite your references. These are the ff. scientists:• Gabriel Fahrenheit• Antoine Lavoisier• James Watt• Benjamin Thompson• Nicolas Sadi Carnot• James Joule• Julius Von Meyer• William Thompson• Rudolf Clausius You can add if there are others. Thank you!arrow_forwardCurrent Attempt in Progress Your answer is partially correct. The p-V diagram in the figure shows two paths along which a sample of gas can be taken from state a to state b, where V, = 3.0V1. Path 1 requires that energy equal to 5.00p,V1 be transferred to the gas as heat. Path 2 requires that energy equal to 6.00p,V1 be transferred to the gas as heat. What is the ratio P2/p1? 2 a 1 9. V V1 Number i 3 Units No unitsarrow_forwardCONCEPTUAL QUESTIONS ON 2ND LAW OF THERMODYNAMICS (10 points) 1. Is a temperature difference necessary to operate a heat engine? State why or why not. 2. Definitions of efficiency vary depending on how energy is being converted. Compare the definitions of efficiency for the human body and heat engines. How does the definition of efficiency in each relate to the type of energy being converted into doing work? 3. Why-other than the fact that the second law of thermodynamics says reversible engines are the most efficient-should heat engines employing reversible processes be more efficient than those employing irreversible processes? Consider that dissipative mechanisms are one cause of irreversibility. PRACTICE PROBLEMS (20 points) 1. (a) What is the efficiency of a cyclical heat engine in which 75.0 kJ of heat transfer occurs to the environment for every 95.0 kJ of heat transfer into the engine? (b) How much work does it produce for 100 kJ of heat transfer into the engine? 2. The engine…arrow_forward
- Introduction to the Ideal Gas Law Learning Goal: To understand the ideal gas law and be able to apply it to a wide variety of situations. The absolute temperature T. volume V, and pressure p of a gas sample are related by the ideal gas law, which states that PV = nRT. Here is the number of moles in the gas sample and R is a gas constant that applies to all gases. This empirical law describes gases well only if they are sufficiently dilute and at a sufficiently high temperature that they are not on the verge of condensing. In applying the ideal gas law, p must be the absolute pressure, measured with respect to vacuum and not with respect to atmospheric pressure, and I must be the absolute temperature, measured in kelvins (that is, with respect to absolute zero, defined throughout this tutorial as -273° C). If p is in pascals and V is in cubic meters, use R = 8.3145 J/(mol-K). If p is in atmospheres and V is in liters, use R=0.08206 L-atm/(mol-K) instead. ▾ ISH P₂ = 1.07P1 Submit Part B…arrow_forwardProblem 1.5. The relatively unique properties of water are among the reasons why it plays such a key role in the Earth's climate. Among those properties are the ones that relate energy and water processes. Provide all answers in standard SI units. a) Define the differences (in meaning) of the heat capacity of a substance and latent heat (i.e. of vaporization or fusion) of a substance. Which one is used in the context of evaporation to convert between a mass flux density and an energy flux density? What are the typical values of these properties for water at 0°C? b) What is the nominal density of liquid water? What is the nominal density of ice? Which is bigger? c) Suppose you have a 2 cm x 2 cm x 2 cm ice cube that is frozen a temperature of 0°C. How much energy input would be required to melt it? d) Suppose you are making yourself 0.50 liters of tea but its temperature is 75°C, which is too hot for you to drink. In order to cool its temperature, you immerse three ice cubes ofarrow_forward1 kg of a gas at a temperature of 40° C and a pressure of 120 Pa occupies a volume of0.25m°. If the Cv of the gas =743 J/Kg.K Find:a.The characteristic gas constantb.The molar mass and relative molecular massC.The specific heat capacity at constant pressured. The ratio of specific heats question is attached please solve showing full working and all formulas used along with all explanations and solve all section also there is a typing error its molar mass not modulararrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

University Physics (14th Edition)
Physics
ISBN:9780133969290
Author:Hugh D. Young, Roger A. Freedman
Publisher:PEARSON

Introduction To Quantum Mechanics
Physics
ISBN:9781107189638
Author:Griffiths, David J., Schroeter, Darrell F.
Publisher:Cambridge University Press

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Lecture- Tutorials for Introductory Astronomy
Physics
ISBN:9780321820464
Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina Brissenden
Publisher:Addison-Wesley

College Physics: A Strategic Approach (4th Editio...
Physics
ISBN:9780134609034
Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart Field
Publisher:PEARSON
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY