
How does the polar character of water molecules help to explain waters relatively large dielectric constant? (Figure 19.19)
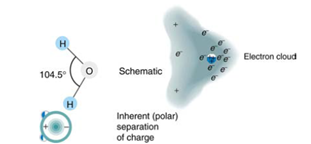
Figure 19,19 Artist's conception of a water molecule. There is an inherent separation of charge, and so water is a polar molecule. Electrons in the molecule are attracted to the oxygen nucleus and leave an excess of positive charge near the two hydrogen nuclai. (Note that the schematic on the right is a rough illustration of the distribution of electrons in the water molecule. It does not show the actual numbers of protons and elections involved in the structure.)
Trending nowThis is a popular solution!

Chapter 19 Solutions
College Physics
Additional Science Textbook Solutions
Essential University Physics: Volume 2 (3rd Edition)
Tutorials in Introductory Physics
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
College Physics: A Strategic Approach (4th Edition)
Physics: Principles with Applications
College Physics: A Strategic Approach (3rd Edition)
- (a) Sketch the electric field pattern around two positive point charges of magnitude 1 C placed close together. (b) Sketch the electric field pattern around two negative point charges of 2 C, placed dose together. (c) Sketch the pattern around two point charges of +1 C and 2 C, placed dose together.arrow_forwardIf qa = 0 in Figure 18-46, under what conditions will there be no net Coulomb force on q?arrow_forwardElaborate on the way in which the polar character of water molecules helps to explain water's relatively large dielectric constant.arrow_forward
- Use the characteristics of the of the Coulomb force to explain why capacitance should be proportional to the plate area of a capacitor. Similarly, explain why capacitance should be inversely proportional to the separation between plates.arrow_forwardA capacitor is designed so that one plate is large and the other is small. If the plates are connected to a battery, (a) the large plate has a greater charge than the small plate, (b) the large plate has less charge than the small plate, or (c) the plates have equal, but opposite, charge.arrow_forwardA 50.0 g ball of copper has a net charge of 2.00 C. What fraction of the copper s electrons has been removed? (Each copper atom has 29 protons, and copper has an atomic mass of 63.5.)arrow_forward
- If the electric field at a point on the line between two charges is zero, what do you know about the charges?arrow_forwardReview. A molecule of DNA (deoxyribonucleic acid) is 2.17m long. The ends of the molecule become singly ionized: negative on one end, positive on the other. The helical molecule acts like a spring and compresses 1.00% upon becoming charged. Determine the effective spring constant of the molecule.arrow_forwardIf 1.801020electrons move through a pocket calculator during a full day’s operation, how many coulombs of charge moved through it?arrow_forward
- Suppose a speck of dust in an electrostatic precipitator has l.0000 1012 protons in it and has a net charge of5.00 nC (a very large charge for a small speck). How many electrons does it have?arrow_forward(a) Sketch the electric field pattern around two positive point charges of magnitude 1 C placed close together. (b) Sketch the electric field pattern around two negative point charges of 2 C, placed dose together. (c) Sketch the pattern around two point charges of +1 C and 2 C, placed dose together.arrow_forwardCommon static electricity involves charges ranging from nanocoulombs to microcoulombs. (a) How many electrons are needed to form a charge of 2.00 nC (b) How many electrons must be removed from a neutral object to leave a net charge of 0.500 C ?arrow_forward
 College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning





