
Concept explainers
(a)
Interpretation:
The product formed in the given reaction is to be stated.
Concept introduction:
Oxime belongs to the family of imines. The formula of oxime is
Answer to Problem 19.48AP
The product formed in the given reaction is cyclohexanone oxime in the presence of hydroxylamine and pyridine. The reaction is shown below.
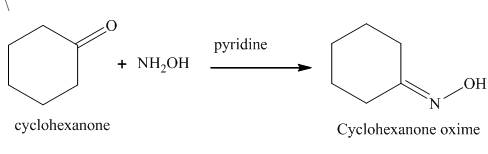
Explanation of Solution
Cyclohexanone reacts with hydroxylamine to form the derivative of the oxime of the carbonyl compound. The nucleophilic addition takes place in the given reaction. The hydroxylamine is used as a nucleophile. The corresponding

Figure 1
The product formed in the given reaction is shown in Figure 1.
(b)
Interpretation:
The product formed in the given reaction is to be stated.
Concept introduction:
The reaction between
Answer to Problem 19.48AP
The product obtained in the given reaction is shown below.
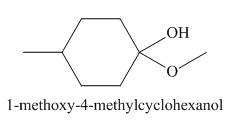
Explanation of Solution
In the given reaction,
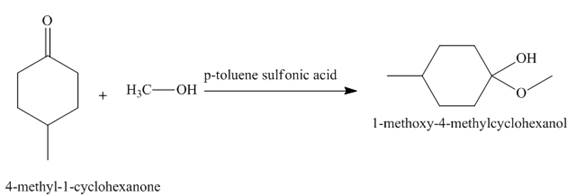
Figure 2
The treatment of
(c)
Interpretation:
The product formed in the given reaction is to be stated.
Concept introduction:
Ketones react with
Answer to Problem 19.48AP
The product formed in the given reaction is a hemiacetal as shown below.
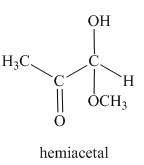
Explanation of Solution
In the third reaction, the methylglyoxal reacts with
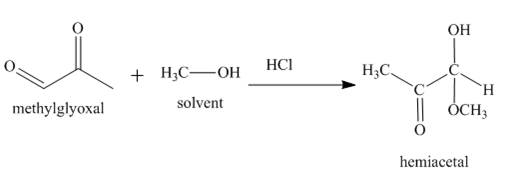
Figure 3
The reaction between methylglyoxal and
(d)
Interpretation:
The product formed in the given reaction is to be stated.
Concept introduction:
The cyclic ethers are ring compounds which contain nitrogen, sulfur and oxygen groups within the ring. The easy method for synthesis of cyclic ether is Williamson synthesis.
Answer to Problem 19.48AP
The product formed when the
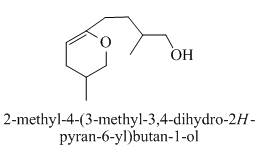
Explanation of Solution
The compound
When acid is treating with
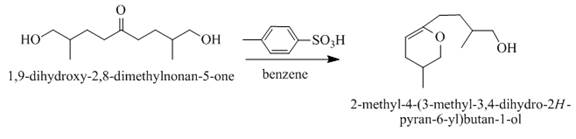
Figure 4
The product formed on the treatment of
(e)
Interpretation:
The product formed in the given reaction is to be stated.
Concept introduction:
Grignard reagents are
Answer to Problem 19.48AP
The compound
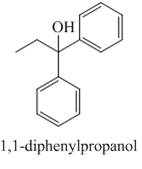
Explanation of Solution
The compound
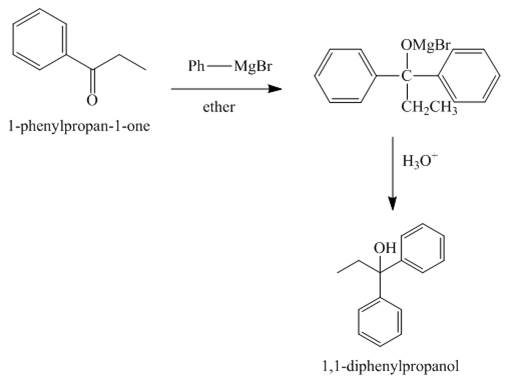
Figure 5
The reaction between
(f)
Interpretation:
The product formed in the given reaction is to be stated.
Concept introduction:
The conversion of an aldehydic or ketonic group to
Answer to Problem 19.48AP
The product formed in the given reaction is
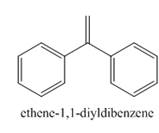
Explanation of Solution
The carbonyl compounds react with Wittig reagent. The product formed is alkenes. In this Wittig reaction, most of the ketone reacts with triphenyl phosphonium ylide to generate an alkene and triphenylphosphine oxide as shown below.
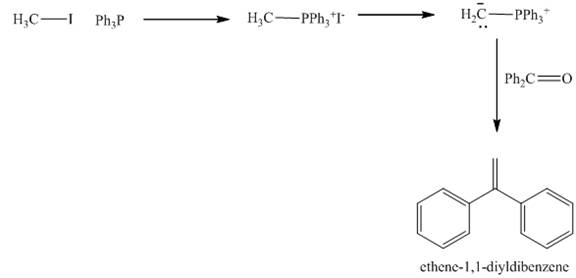
Figure 6
The treatment of haloalkane with triphenylphosphine gives the product
(g)
Interpretation:
The product formed in the given reaction is to be stated.
Concept introduction:
The Wittig reaction is the chemical reaction involves a change in an aldehyde or ketone converted into an alkene. The chemical name of Wittig reagent is triphenyl phosphonium ylide. The Wittig reagent gives good yields of alkene even when other functional groups are present on the aldehydes or ketone. However, as the steric hindrance of the aldehydes or ketone increases, the yield of alkene decreases.
Answer to Problem 19.48AP
The product formed in the given reaction is shown below.
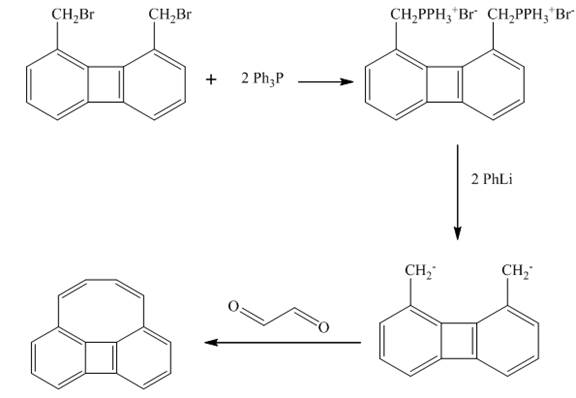
Explanation of Solution
The reaction is Wittig reaction. The reagent used is is triphenyl phosphonium ylide.
In the first step, the treatment of triphenyl phosphonium ylide reacts with

Figure 7
The given reaction is shown in Figure 7 in the presence of Wittig reagent.
Want to see more full solutions like this?
Chapter 19 Solutions
Loose-leaf Version For Organic Chemistry
- Give a clear handwritten answer with textual explanation needed...give the mechanism of given bleow reactionarrow_forwardGive a clear handwritten answer...complete the following reactions with detailed explanation neededarrow_forwardBased on the theoretical discussion, illustrate with equations: a. How ethers are broken down by zinc chloridearrow_forward
- Compound F may be synthesised by the method attached Draw the isomer of compound B and explain which one would be the major product and why.arrow_forwardthe following reaction scheme leads to the formation of compound C. give the structure of the final product C and of the intermediate products A and B and justify, using the mechanism, the formation of the product A. Give the serereochemistry of the final product obtainedarrow_forwardExplain clearly Giving and example and mechanisms, explain the aromatic Substitution reactions.arrow_forward
- Give a clear handwritten answer with explanation needed...give the mechanism of given bleow reactionarrow_forwardGive the structural chemistry of active methylene, classify it asradical/intermediate/stable organic compound, reagent of its production and role in alkylationarrow_forwardin part b. isomer of compound B. Which one would be the major product and why?arrow_forward
- the following reaction scheme leads to the formation of compound B. give the structure of the final products and of the intermediate product A and justify, using the mechanism, the formation of thesearrow_forwardGive detailed Solution...Draw the following organic compoundarrow_forwardCompound A was oxidized with periodic acid to give B, which after acid hydrolysis gave C. Bromine oxidation of C gave D. Suggest structural formulas, including stereochemistry, for compounds B, C, and D.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





