
Concept explainers
(a)
Interpretation:
The skeletal structure of
Concept Introduction:
Fatty acids are long-chain
Unsaturated fatty acids can be defined as the long-chain fatty acids which have a long hydrocarbon chain with −COOH group. In unsaturated fatty acids, the carbon chain must have at least one double bond.
Answer to Problem 34P
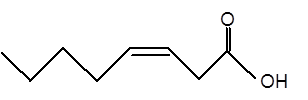
Explanation of Solution
Given:
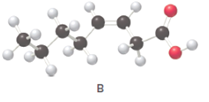
Fatty acids are long-chain carboxylic acids, which consist of two parts; long hydrocarbon chains and polar −COOH group. The long hydrocarbon chain consists of carbon and H atoms, so it has C-C and C-H bonds only, whereas, the polar −COOH group has polar C=O and C-O bonds. The unsaturated fatty acids have cis and trans-double bonds in the hydrocarbon chain.
In the ball-and-stick model, the red ball represents O atom, the black ball represents C atom and the white ball represents H atoms. Thus, the skeletal structure is:
(b)
Interpretation:
The omega-n designation for the
Concept Introduction:
Fatty acids are long-chain carboxylic acid, which may or may not have unsaturation in the molecule. They react with glycerol to form triglycerides. The reaction is called esterification as a carboxylic acid group reacts with the alcoholic group to form an ester group.
Unsaturated fatty acids can be defined as the long-chain fatty acids which have a long hydrocarbon chain with −COOH group. In unsaturated fatty acids, the carbon chain must have at least one double bond.
Answer to Problem 34P
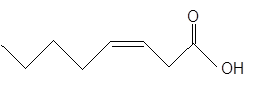
The
Explanation of Solution
Fatty acids are long-chain carboxylic acids, which consist of two parts; long hydrocarbon chains and polar −COOH group. If there is at least one double bond in the long hydrocarbon chain of the fatty acid, it is said to be an unsaturated fatty acid.
The unsaturated fatty acids can also classify as omega-n acids. Here, 'n' represents the position of the first
In the given fatty acid
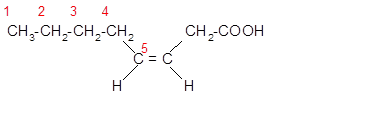
(c)
Interpretation:
The stereoisomer of the
Concept Introduction:
Fatty acids are long-chain carboxylic acid, which may or may not have unsaturation in the molecule. They react with glycerol to form triglycerides. The reaction is called esterification as a carboxylic acid group reacts with the alcoholic group to form an ester group.
Unsaturated fatty acids can be defined as the long-chain fatty acids which have a long hydrocarbon chain with −COOH group. In unsaturated fatty acids, the carbon chain must have at least one double bond.
Answer to Problem 34P
The stereoisomer of cis-fatty acid must be trans-isomer as given below;
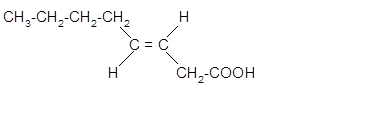
Explanation of Solution
Fatty acids are long-chain carboxylic acids, which consist of two parts; long hydrocarbon chains and polar −COOH group. The double bond can arrange in two ways, i.e., cis and Trans in the hydrocarbon chain of the fatty acid molecule. Hence, the stereoisomer of cis-fatty acid
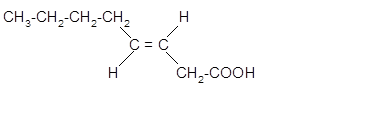
(d)
Interpretation:
The structure of wax formed by the reaction of
Concept Introduction:
Fatty acids are long-chain carboxylic acid, which may or may not have unsaturation in the molecule. They react with glycerol to form triglycerides. The reaction is called esterification as a carboxylic acid group reacts with the alcoholic group to form an ester group.
Unsaturated fatty acids can be defined as the long-chain fatty acids which have a long hydrocarbon chain with −COOH group. In unsaturated fatty acids, the carbon chain must have at least one double bond.
Answer to Problem 34P
The skeleton formula of wax formed by the reaction of
Explanation of Solution
Fatty acids are long-chain carboxylic acids, which consist of two parts; long hydrocarbon chains and polar −COOH group. Waxes are a good example of hydrolyzable lipids, which are composed of fatty acid and higher alcohols. They have an ester
Hence, the reaction of
Want to see more full solutions like this?
Chapter 19 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
- 1. Assign a name to each of the following monosaccharides. Use D and L designations. H I CH 2OH C=O C=O a. b. a. HO-C-H H-C-OH H-C-OH CH₂OH d. True or False: b. HỌCH CH ₂OH C. Circle all chiral centers for the monosaccharides shown. The molecules shown can rotate plane polarized light?arrow_forward33. Determine whether or not each molecule is a fatty acid. If it is a fatty acid, classify it as saturated, monounsaturated, or polyun- saturated. a. CH HO, b. CH, CH CH, . CH CH;-C-OH d. CH, CH=CHarrow_forwardWhich of the following types of carbohydrates can be hydrolyzed to give smaller molecules? I. monosaccharides II. disaccharides II. oligosaccharides IV. polysaccharides 7. a.l only b.Il and IV only c. II only d.l and II only e. II, III, and IVarrow_forward
- 3. Lipids a. What are saturated fats? b. What is the main origin of triacylglycerides or oils? O c. Why do unsaturated oils discolor bromine in solution? d. What functional groups are present in triacylglycerides? and. Why are lipids not soluble in water when they have a polar part? F. What positive function does cholesterol have in the body? pility: Good to go D (Ctrl)arrow_forwardGlycogen is more rigid than starch even though both are made up of D-glucose monomers. What could be the reason for this phenomenon? Select one: а. Starch has less (al→4) linkages b. Glycogen has more branching (al→6) linkages c. Glycogen has more (B1→4) linkages to allow for stronger H-bond interactions. d. Starch has more branching (al→6) linkages e. Glycogen has more glucose unitsarrow_forwardV F2 # 3 E 80 F3 What is the difference between integral and peripheral proteins? $ 4 F4 R A) Integral proteins are required for the cell to function, while peripheral proteins are accessory. B) Integral proteins are found on the outside of the cell, while peripheral proteins are found on the inside of the cell. C) Integral proteins extend through the lipid bilayer, and peripheral proteins are just on the surface of the inside or outside of the cell. Question 11 of 19 D) Integral proteins are found on the inside of the cell, while peripheral proteins are found on the outside of the cell. E) Integral proteins are channel proteins and peripheral proteins act as enzymes. % 5 F5 T <6 MacBook Air F6 Y & 7 ♡ F7 U * 8 DII F8 1 ( - 9 F9 0 O F10arrow_forward
- Which fatty acid would you expect to be have the lowest melting point? # 3 80 F3 71 $ 4 A) CH,CH=CH(CH,), CH=CHCOOH B) CH₂(CH₂),COOH C) CH₂(CH₂),COOH D) CH₂CH=CH(CH₂),COOH E) CH₂(CH₂) ₁4COOH Q F4 dº L % 5 Question 6 of 19 F5 14 <6 F6 & 7 ▷▷ F7 * 0 8 DII F8 9 DD F9 0 7 F10arrow_forward1. HO HO OH Monosaccharide 2. Disaccharide H HO Polysaccharide H. OH 3. Starcharrow_forwardb eing no 2. Palmitoleic acid is a fatty acid with the following condensed structural formula: O || CH3-(CH₂)5-CH=CH-(CH₂)7-C-OH a. How many carbon atoms are in palmitoleic acid? b. Is the fatty acid saturated, monounsaturated, or polyunsaturated? c. Is it most likely to be solid or liquid at room temperature?arrow_forward
- 12. Monosaccharides are designated with a "D" or "L" in their name. What determines this? A. The bond between a chiral carbon and an OH group in the structure. B. The acyclic monosaccharide contains either a ketone or an aldehyde functional group. C. The molecule has either five or six carbons in its structure. D. The molecule is either cyclic or acyclic. E. The anomeric carbon has an OH group pointing up or down in a drawing of the molecule.arrow_forwardA glycosidic bond connects two monosaccharides to create sucrose (more commonly known as table sugar). Identify the type of glycosidic bond connecting the monosaccharides together as well as these two monosaccharides that are found in sucrose. a. α,ß-1,2-glycosidic bond, ß-L-glucose, and α- L-fructose b. α,ß-1,2-glycosidic bond, α-D-galactose, and ß-D-glucose c. α-1,4-glycosidic bond, α-D-glucose, and ß-D-fructose d. α,ß-1,2-glycosidic bond, α-D-glucose, and ß-D-fructosearrow_forward16. How does a double bond in a fatty acid change the overall structure of the fatty acid? а. The overall structure does not change. b. The more double bonds a fatty acid has, the more linear it is. The more double bonds a fatty acid has, the more flexible it is. d. The more double bonds a fatty acid has, the more bent or kinked it is. С. e. The more double bonds a fatty acid has, the more twisted it is. 17. A polyunsaturated fatty acid contains multiple ketones. carboxylic acids. carbon-carbon double bonds. alcohols. Any of the above ing the deltaarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





