
Concept explainers
Answer the following questions about the given triacylglycerol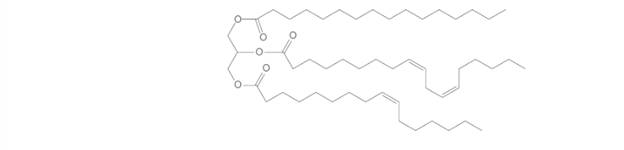
a. What fatty acids are used to form this triacylglycerol?
b. Would you expect this triacylglycerol to be a solid or a liquid at room temperature?
c. What regions are hydrophobic?
d. What regions are hydrophilic?
e. What hydrolysis products are formed when the triacylglycerol is treated with aqueous sulfuric acid?
(a)
Interpretation:
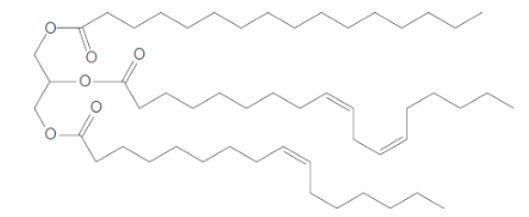
The fatty acids which are used to form the below triacylglycerol needs to be determined.
Concept Introduction:
Lipids are biomolecules that are involved in different biochemical reactions. They are special types of organic molecules which can only be identified with the help of their physical properties, not by the presence of any certain functional group. In general, lipids contain a large number of C-C and C-H bonds with few polar functional groups such as −OH, -SH, etc. For example, triacylglycerol consists of three ester groups whereas Vitamin E is composed of both ether and phenolic groups.
Answer to Problem 50P
In the given triacylglycerol; the fatty acids must be:
Explanation of Solution
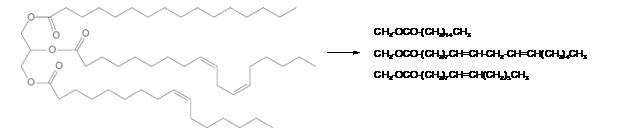
Triacylglycerols are most abundant lipids which are mainly found in animal fat and vegetable oils. They are triesters of glycerol, therefore in the formation of one molecule of triacylglycerol, three molecules of fatty acids react with one molecule of glycerol as given below;
Hence the left side part of hydrocarbon chains is part of fatty acids in the triacylglycerol molecule. The structural formula for the given triacylglycerol is as given below.
In the given triacylglycerol; the fatty acids must be:
(b)
Interpretation:
The physical state of given triacylglycerol needs to be determined.
Concept Introduction:
Lipids are biomolecules that are involved in different biochemical reactions. They are special types of organic molecules which can only be identified with the help of their physical properties, not by the presence of any certain functional group. In general, lipids contain a large number of C-C and C-H bonds with few polar functional groups such as −OH, -SH, etc. For example, triacylglycerol consists of three ester groups whereas Vitamin E is composed of both ether and phenolic groups.
Answer to Problem 50P
The triacylglycerol must be in a liquid state.
Explanation of Solution
In the given triacylglycerol; the fatty acids must be:
Linoleic acid and Palmitoleic acid are unsaturated fatty acids as they have C=C bonds in the long hydrocarbon chain. The unsaturation in fatty acids decreases the melting point of molecule due to bending in the molecule. Since the given triacylglycerol is composed of unsaturated fatty acids, therefore, it should exist in a liquid state.
(c)
Interpretation:
The hydrophobic part of given triacylglycerol needs to be determined.
Concept Introduction:
Lipids are biomolecules that are involved in different biochemical reactions. They are special types of organic molecules which can only be identified with the help of their physical properties, not by the presence of any certain functional group. In general, lipids contain a large number of C-C and C-H bonds with few polar functional groups such as −OH, -SH, etc. For example, triacylglycerol consists of three ester groups whereas Vitamin E is composed of both ether and phenolic groups.
Answer to Problem 50P
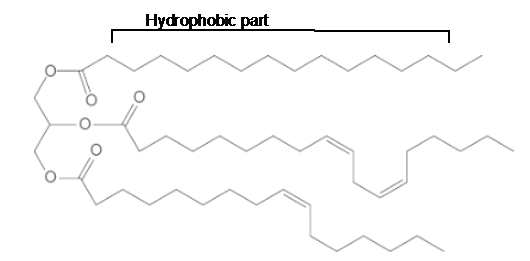
The long hydrocarbon chains in the triacylglycerol molecule is hydrophobic part of the molecule.
Explanation of Solution
Triacylglycerols are most abundant lipids which are mainly found in animal fat and vegetable oils. They are triesters of glycerol, therefore in the formation of one molecule of triacylglycerol, three molecules of fatty acids react with one molecule of glycerol as given below;
The long hydrocarbon chains of the triacylglycerol are hydrophobic part as it is non-polar part.
(d)
Interpretation:
The hydrophilic part of given triacylglycerol needs to be determined.
Concept Introduction:
Lipids are biomolecules that are involved in different biochemical reactions. They are special types of organic molecules which can only be identified with the help of their physical properties, not by the presence of any certain functional group. In general, lipids contain a large number of C-C and C-H bonds with few polar functional groups such as −OH, -SH, etc. For example, triacylglycerol consists of three ester groups whereas Vitamin E is composed of both ether and phenolic groups.
Answer to Problem 50P
The ester linkage in the triacylglycerol molecule is hydrophilic part of the molecule.
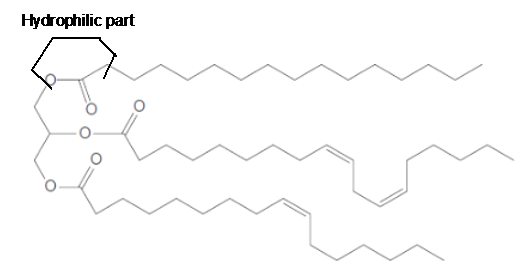
Explanation of Solution
Triacylglycerols are most abundant lipids which are mainly found in animal fat and vegetable oils. They are triesters of glycerol, therefore in the formation of one molecule of triacylglycerol, three molecules of fatty acids react with one molecule of glycerol as given below;
The long hydrocarbon chains of the triacylglycerol are hydrophobic part as it is non-polar part whereas the ester linkage in the molecule is hydrophilic part of the molecule.
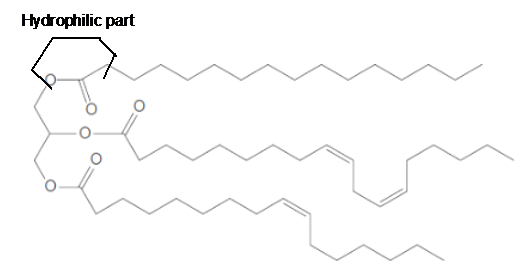
(e)
Interpretation:
The hydrolysis product of given triacylglycerol with aqueous sulfuric acid needs to be determined.
Concept Introduction:
Lipids are biomolecules that are involved in different biochemical reactions. They are special types of organic molecules which can only be identified with the help of their physical properties, not by the presence of any certain functional group. In general, lipids contain a large number of C-C and C-H bonds with few polar functional groups such as −OH, -SH, etc. For example, triacylglycerol consists of three ester groups whereas Vitamin E is composed of both ether and phenolic groups.
Answer to Problem 50P
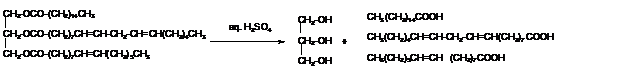
Explanation of Solution
Triacylglycerols are most abundant lipids which are mainly found in animal fat and vegetable oils. They are triesters of glycerol, therefore in the formation of one molecule of triacylglycerol, three molecules of fatty acids react with one molecule of glycerol as given below;
The hydrolysis of triacylglycerol with aqueous sulfuric acid leads to the formation of fatty acid and glycerol molecule.
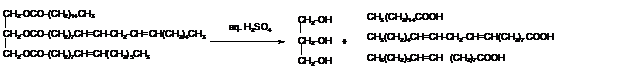
Want to see more full solutions like this?
Chapter 19 Solutions
Student Study Guide/solutions Manual To Accompany Format: Paperback
- Identify each of the following characteristics as describing an unsaturated fatty acid or a saturated fatty acid: a. Contains more hydrogen atoms b. Is more healthy c. More plentiful in plant sources d. Is ususally a solid at room temperaturearrow_forwardThe plasmalogens are a group of lipids found in nerve and muscle cells. How do plasmalogens differ from fats?arrow_forwardDraw structures of glyceryl tripalmitate and glyceryl trioleate. Which would you expect to have a higher melting point?arrow_forward
- 4. Which of the following best describe the physical properties of fatty acids?a. All physical properties of fatty acids are influenced by the length and degreeof unsaturation of their carbon chain.b. Melting points of fatty acids are influenced by the carbon chain length.c. Melting points of fatty acids are influenced by their degree of unsaturation.d. Alltheabove.5. Triacylglycerols are ________.a. insolubleinwater.b. partially soluble in water.c. soluble in water at elevated temperatures.d. solubleinwater.6. The backbone of a triglyceride is __________.a. a three-carbon molecule with an aldehyde and two hydroxyl groups.b. a three-carbon molecule with three hydroxyl groups.c. esterified fatty acids fused with 1 to 3 carbons.d. threelong-chainfattyacids.7. Triacylglycerols are _____ in their ability to store energy when compared to glycogen, the energy storing form of glucose in the human body.a. equally efficientb. insignificantc. less efficientd. more efficient 8. The essential…arrow_forwardA triglyceride can be optically active if it contains two or more different fatty acids.(a) Draw the structure of an optically active triglyceride containing one equivalent of myristic acid and two equivalentsof oleic acidarrow_forwardWhat is the difference between saturated fatty acids and unsaturated fatty acids? Pick one answer. A. Unsaturated fatty acids have an even number of carbons, saturated fatty acids have an odd number of carbons. B. Unsaturated fatty acids have cis double bonds, saturated fatty acids have trans double bonds. C. Unsaturated fatty acids have double bonds, saturated fatty acids do not. D. Unsaturated fatty acids are naturally occurring compounds, saturated fatty acids do not occur in nature and must be prepared synthetically.arrow_forward
- please provide additional info regarding this "A saturated fatty acid sample has a high boiling point. A triacylglycerol is composed of glycerol and three saturated fatty acids. Therefore, a sample not containing this triacylglycerol will have a low boiling point."arrow_forwardHow would you expect the melting points of the following unsaturated fatty acids to compare? Explain your choice.arrow_forwardExplain the Most Common Fatty Acids in Triacylglycerols ?arrow_forward
- Why might some food companies find it economically advantageous to advertise their products (such as corn oil and margarine) as being composed of polyunsaturated fatty acids?arrow_forward1. Draw, using condensed structural formulas, all reactants and products of the saponification reaction of a triacylglycerol using NaOH. Use lauric acid, a 12-carbon saturated fatty acid found in vegetable shortening. 2. Soap can be made using a variety of different fats, oils and bases. Suppose a student wanted to make a soft, liquid soap that lathers easily. What type(s) of fat/oil and base would you recommend? How would this differ from making a solid bar of soap? 3. Vegetable oils are typically liquid at room temperature, yet Crisco (which is made of vegetable oil) is solid at room temperature. Explain why/how this is the case. 4. Explain, with the use of a diagram how soap molecules work to clean “dirt” particles. 5. Amylase is a natural enzyme found in saliva. Describe this enzyme’s function. 6. Lactose is a sugar found in milk and other dairy products. Explain how it is broken down in most individuals. Explain lactose “intolerance”.arrow_forward14. Which lipid does not contain any fatty acids?A.GlycerophospholipidB. triglycerideC. steroidD. wax E. sphingolipidarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





