
EBK A SMALL SCALE APPROACH TO ORGANIC L
4th Edition
ISBN: 9781305446021
Author: Lampman
Publisher: CENGAGE LEARNING - CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 19, Problem 6Q
Interpretation Introduction
Interpretation:
The behavior of given compounds in the silver nitrate test and sodium iodide test needs to be explained.
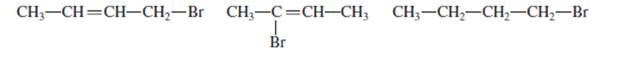 Concept Introduction:
Concept Introduction:
Here HX can be HCl or
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
10. What is the order of increase in acidity for the following compounds?
Explain how benzaldehyde and dimedone reacts with each other, and then with the aminotriazole to form compound 1a in the presence of an acid catalyst. Provide a detailed reaction mechanism.
During the development of the optimized procedure for the experiment, it was found out that compound 1b can also be produced from the same set of starting materials. Propose a detailed reaction mechanism for the formation of 1b. Explain your answer.
What factor/s may drive the formation of 1b over 1a?
Following is a 1H-NMR spectrum of2-butanol, Explain why the CH2 protons appear as a complex multiplet rather than as a simple quintet.
Chapter 19 Solutions
EBK A SMALL SCALE APPROACH TO ORGANIC L
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 9. I did a TLC analysis of benzophenone and triphenylmethanol. I had expected the triphenylmethanol to have a lower Rf value being the more polar amongst the two compounds, but the reverse is the case. I find it difficult to rationalize this observation. Can I get an explanation please?arrow_forwardWhy must the stopper at the top of the separatory funnel be removed before liquid can be withdrawn through the stopcock? - Write four equations (showing structures) for the reactions used in this experiment to separate and retrieve \ ( \mathrm {p} \)-bromoaniline and benzoic acid.arrow_forwardWhat changes in NMR and IR spectra would you expect to see upon acylation of benzene? Explicitly discuss the elements of the 1H NMR and IR of acetophenone that you would use to argue that the reaction was successful.arrow_forward
- The azo compound shown below has a λmax value of 410 nm. When treated with hydrogen in the presence of palladium on carbon, the λmax changes significantly. Suggest a reason for this observation and predict whether the λmax value increases or decreases.arrow_forwardAfter the reduction of camphor with NaBH4 experiment, you took a 1H-NMR of your product sample. The sample ave the following 1H-NMR singlas with the given integrations (2.314 and 9.497). Determine the product ratio of isoboreneol to borneol in this particular mixture.arrow_forwardHow does the proton (1H) NMR of the methyl benzoate reagent compare to the methyl 3-nitrobenzoate product? Include (or illustrate) the NMR plots and peak assignments for each please type out so i can read clearlyarrow_forward
- What products would you would expect to obtain when the following compounds react with ozone and then with dimethylsulfide?arrow_forwardThe 1H-NMR spectrum of lycopene is provided below. Comment on the general features of the NMR spectrum.arrow_forwardDiscuss the similarities and difference you expect to see in their FT-IR spectra by going from compound 1 to 2arrow_forward
- Propose structures for compounds that fit the following data: (a) A ketone with M+=86 and fragments at m/z=71 and m/z=43 (b) An alcohol with M+=88 and fragments at m/z=73, m/z=70, and m/z=59arrow_forwardDraw the structure of the compound that produced the spectra below. The infrared spectrum has strong bands at 1720 and 1738 cm-1.arrow_forwardProvide a detailed analysis of IR, Mass, 1H NMR and 13C NMR data for the following compound.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY