
Organic Chemistry - Access
7th Edition
ISBN: 9780321820020
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19, Problem 82P
- a. Describe four ways the following reaction can be earned out:
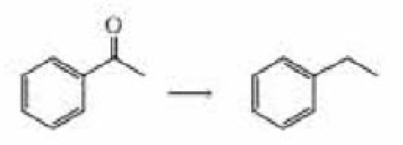
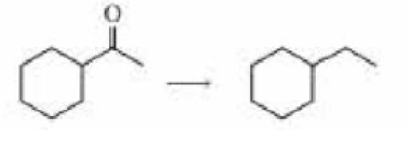
- b. Describe three ways the following reaction can be carried out:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(a) Explain why an alkylamine is more basic than ammonia?(b) How would you convert(i) Aniline to nitrobenzene (ii) Aniline to iodobenzene
The name for Reaction 1 is _____ while Reaction 2 is called _____.
Choices: A. Williamson Ether synthesis, B. Hydration, C. Epoxidation, D. Acidic cleavage
The reagent/s for REACTION 1 is/are ______
Choices: A. m-chloroperoxybenzoic acid, B. H2O/H3PO4, C. H3O+, D. HI
The reagent/s for REACTION 2 is/are ______
Choices: A. m-chloroperoxybenzoic acid, B. water in acidic medium, C. dilute acid, D. hydroiodic acid
The reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, doesnot work with primary alcohols.
a. Why does the Ritter reaction not work with primary alcohols?
b. Provide an explanation for why an amide is less susceptible to nucleophilic attack than its corresponding ester.
Chapter 19 Solutions
Organic Chemistry - Access
Ch. 19.1 - Draw the structure for each of the following: a....Ch. 19.2 - PROBLEM 2
If electrophilic addition to benzene is...Ch. 19.4 - Why does hydration inactivate FeBr3?Ch. 19.6 - Prob. 5PCh. 19.7 - Prob. 6PCh. 19.8 - What is the major product of a Friedel-Crafts...Ch. 19.10 - Describe two ways to prepare each of the following...Ch. 19.12 - Prob. 9PCh. 19.13 - Name the following:Ch. 19.13 - Prob. 12P
Ch. 19.13 - Prob. 13PCh. 19.13 - Prob. 14PCh. 19.14 - Prob. 15PCh. 19.14 - List the compounds in each set from most reactive...Ch. 19.15 - Prob. 18PCh. 19.15 - What product(s) result from nitration of each of...Ch. 19.15 - Prob. 20PCh. 19.16 - Which acid in each of the following pairs is...Ch. 19.16 - Prob. 23PCh. 19.16 - Prob. 24PCh. 19.18 - Show how the following compounds can be...Ch. 19.18 - Give the products, if any, of each of the...Ch. 19.19 - a. Does a coupling reaction have to be used to...Ch. 19.19 - PROBLEM 28
Show how each of the following...Ch. 19.20 - What is the major product(s) of each of the...Ch. 19.20 - Prob. 30PCh. 19.21 - Why isn't FeBr3 used as a catalyst in the first...Ch. 19.21 - Prob. 33PCh. 19.21 - Write the sequence of steps required for the...Ch. 19.21 - Prob. 35PCh. 19.22 - What product is formed from reaction of...Ch. 19.22 - Prob. 37PCh. 19.22 - Draw the structure of the activated ring and the...Ch. 19.23 - Prob. 39PCh. 19.23 - Prob. 40PCh. 19.23 - Diazomethane can be used to convert a carboxylic...Ch. 19.24 - Prob. 42PCh. 19.24 - Prob. 43PCh. 19.24 - Prob. 44PCh. 19.25 - Prob. 45PCh. 19 - Draw the structure for each of the following: a....Ch. 19 - Prob. 47PCh. 19 - Prob. 48PCh. 19 - Prob. 49PCh. 19 - For each of the statements in Column I, choose a...Ch. 19 - What product is obtained from the reaction of...Ch. 19 - Draw the product(s) of each of the following...Ch. 19 - Rank the following substituted anilines from most...Ch. 19 - Prob. 54PCh. 19 - The compound with the 1H NMR spectrum shown below...Ch. 19 - Prob. 56PCh. 19 - Show how the following compounds can be...Ch. 19 - Prob. 58PCh. 19 - Rank each group of compounds from most reactive to...Ch. 19 - Prob. 60PCh. 19 - Describe two ways to prepare anisole from benzene.Ch. 19 - For each of the following components, indicate the...Ch. 19 - Prob. 63PCh. 19 - Prob. 64PCh. 19 - Prob. 65PCh. 19 - Prob. 66PCh. 19 - An aromatic hydrocarbon with a molecular formula...Ch. 19 - The following tertiary alkyl bromides undergo an...Ch. 19 - Show how the following compounds can be...Ch. 19 - Use the four compounds shown below to answer the...Ch. 19 - a. Rank the following esters from most reactive to...Ch. 19 - A mixture of 0.10 mol benzene and 0.10 mol...Ch. 19 - Prob. 73PCh. 19 - Benzene underwent a Friedel-Crafts acylation...Ch. 19 - Prob. 75PCh. 19 - Prob. 76PCh. 19 - Prob. 77PCh. 19 - Friedel-Crafts alkylations can be carried out with...Ch. 19 - Show how the following compounds can be prepared...Ch. 19 - Prob. 80PCh. 19 - Prob. 81PCh. 19 - a. Describe four ways the following reaction can...Ch. 19 - Propose a mechanism for each of the following...Ch. 19 - Prob. 84PCh. 19 - Describe how naphthalene can he prepared from the...Ch. 19 - Prob. 86PCh. 19 - Using resonance contributors for the carbocation...Ch. 19 - Prob. 88PCh. 19 - When heated with chromic acid, compound A forms...Ch. 19 - Prob. 90PCh. 19 - Prob. 91PCh. 19 - What reagents are required to carry out the...Ch. 19 - Show how the following compounds can be prepared...Ch. 19 - An unknown compound reacts with ethyl chloride and...Ch. 19 - How can you distinguish the following compounds...Ch. 19 - P-Fluoronitrobenzene is more reactive toward...Ch. 19 - a. Explain why the following reaction leads to the...Ch. 19 - Describe how mescaline can be synthesized from...Ch. 19 - Propose a mechanism for the following reaction...Ch. 19 - Explain why hydroxide ion catalyzes the reaction...Ch. 19 - Propose a mechanism for each of the following...Ch. 19 - Prob. 102PCh. 19 - Prob. 103PCh. 19 - Describe how 3-methyl-1-phenyl-3-pentanol can he...Ch. 19 - a. How can aspirin be synthesized from benzene? b....Ch. 19 - Prob. 106PCh. 19 - Show how Novocain, a painkiller used frequently by...Ch. 19 - Prob. 108PCh. 19 - Saccharin, an artificial sweetener, is about 300...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Nicotinic acid, more commonly named niacin, is one of the B vitamins. Show how nicotinic acid can be converted to (a) ethyl nicotinate and then to (b) nicotinamide.arrow_forward10.14 Complete the following reactions: Show the step-by-step process. Do not use shortcut methods. Make it as detailed as it can be. Encode (not hand-written)!arrow_forwardWhat is the product of the reaction below? Noting that it first reacts with A and then followed by B.arrow_forward
- Finished A-C need help with the remaining D-Iarrow_forwardShow how m-dibromobenzene can be synthesized from benzene from multiple reactions. Tip: think of diazotation as an important reaction in this process.arrow_forwardAnswer each part for the reaction below. a. Draw in ALL missing lone pairs b. Draw appropriate curved arrows to complete the acid-base reaction c. Draw the corresponding products for the reactionarrow_forward
- Can you explain how they got the minor product, show the movement of electrons etc.arrow_forwarda. Draw the structures of all the elimination products. b. The reaction occurs through an E2 mechanism. Using one of your products writethe mechanism of the reaction using curved arrows. c. Is this reaction regioselective? Explain your answer. d. Explain why the reaction is stereoselective.arrow_forwardThe following reaction takes place several times faster than the reaction of 2-chlorobutane with HO-: a. Explain the enhanced reaction rate.b. Explain why the OH group in the product is not bonded to the carbon that was bonded to the Cl group in the reactant.arrow_forward
- (a) Illustrate the following name reactions giving suitable example in each case :(i) Clemmensen reduction (ii) Hell-Volhard-Zelinsky reaction(b) How are the following conversions carried out?(i) Ethylcyanide to ethanoic acid (ii) Butan-l-ol to butanoic acid(iii) Benzoic acid to m-bromobenzoic acidarrow_forwardRank the following according to increasing reactivity with 1 as the least reactive, and 4 as the most reactive in a nucleophilic acyl substitution reaction. Briefly explain your answer.arrow_forwardDraw the final product formed in the reaction below.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY