
Organic Chemistry - MasteringChemistry
7th Edition
ISBN: 9780321867148
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19, Problem 99P
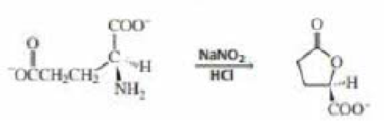
Propose a mechanism for the following reaction that explains why the configuration of the asymmetric center in the reactant is retained in the product:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Understand the structure and sketch the resultant 2 products and their relationship (stereochemistry) with one another and identify their stereocenters (R/S).
Select the necessary reagent(s) for the following transformation.
a. K2Cr2O7
b. NaOH
c. I2
d. Cu^2+
e. H+/H2O
f. H2/Pt
When (R)-6-bromo-2,6-dimethylnonane is dissolved in CH3OH, nucleophilic substitution yields an optically inactive solution. When the isomeric halide (R)-2-bromo-2,5-dimethylnonane is dissolved in CH3OH under the same conditions, nucleophilic substitution forms an optically active solution. Draw the products formed in each reaction, and explain why the difference in optical activity is observed.
Chapter 19 Solutions
Organic Chemistry - MasteringChemistry
Ch. 19.1 - Draw the structure for each of the following: a....Ch. 19.2 - PROBLEM 2
If electrophilic addition to benzene is...Ch. 19.4 - Why does hydration inactivate FeBr3?Ch. 19.6 - Prob. 5PCh. 19.7 - Prob. 6PCh. 19.8 - What is the major product of a Friedel-Crafts...Ch. 19.10 - Describe two ways to prepare each of the following...Ch. 19.12 - Prob. 9PCh. 19.13 - Name the following:Ch. 19.13 - Prob. 12P
Ch. 19.13 - Prob. 13PCh. 19.13 - Prob. 14PCh. 19.14 - Prob. 15PCh. 19.14 - List the compounds in each set from most reactive...Ch. 19.15 - Prob. 18PCh. 19.15 - What product(s) result from nitration of each of...Ch. 19.15 - Prob. 20PCh. 19.16 - Which acid in each of the following pairs is...Ch. 19.16 - Prob. 23PCh. 19.16 - Prob. 24PCh. 19.18 - Show how the following compounds can be...Ch. 19.18 - Give the products, if any, of each of the...Ch. 19.19 - a. Does a coupling reaction have to be used to...Ch. 19.19 - PROBLEM 28
Show how each of the following...Ch. 19.20 - What is the major product(s) of each of the...Ch. 19.20 - Prob. 30PCh. 19.21 - Why isn't FeBr3 used as a catalyst in the first...Ch. 19.21 - Prob. 33PCh. 19.21 - Write the sequence of steps required for the...Ch. 19.21 - Prob. 35PCh. 19.22 - What product is formed from reaction of...Ch. 19.22 - Prob. 37PCh. 19.22 - Draw the structure of the activated ring and the...Ch. 19.23 - Prob. 39PCh. 19.23 - Prob. 40PCh. 19.23 - Diazomethane can be used to convert a carboxylic...Ch. 19.24 - Prob. 42PCh. 19.24 - Prob. 43PCh. 19.24 - Prob. 44PCh. 19.25 - Prob. 45PCh. 19 - Draw the structure for each of the following: a....Ch. 19 - Prob. 47PCh. 19 - Prob. 48PCh. 19 - Prob. 49PCh. 19 - For each of the statements in Column I, choose a...Ch. 19 - What product is obtained from the reaction of...Ch. 19 - Draw the product(s) of each of the following...Ch. 19 - Rank the following substituted anilines from most...Ch. 19 - Prob. 54PCh. 19 - The compound with the 1H NMR spectrum shown below...Ch. 19 - Prob. 56PCh. 19 - Show how the following compounds can be...Ch. 19 - Prob. 58PCh. 19 - Rank each group of compounds from most reactive to...Ch. 19 - Prob. 60PCh. 19 - Describe two ways to prepare anisole from benzene.Ch. 19 - For each of the following components, indicate the...Ch. 19 - Prob. 63PCh. 19 - Prob. 64PCh. 19 - Prob. 65PCh. 19 - Prob. 66PCh. 19 - An aromatic hydrocarbon with a molecular formula...Ch. 19 - The following tertiary alkyl bromides undergo an...Ch. 19 - Show how the following compounds can be...Ch. 19 - Use the four compounds shown below to answer the...Ch. 19 - a. Rank the following esters from most reactive to...Ch. 19 - A mixture of 0.10 mol benzene and 0.10 mol...Ch. 19 - Prob. 73PCh. 19 - Benzene underwent a Friedel-Crafts acylation...Ch. 19 - Prob. 75PCh. 19 - Prob. 76PCh. 19 - Prob. 77PCh. 19 - Friedel-Crafts alkylations can be carried out with...Ch. 19 - Show how the following compounds can be prepared...Ch. 19 - Prob. 80PCh. 19 - Prob. 81PCh. 19 - a. Describe four ways the following reaction can...Ch. 19 - Propose a mechanism for each of the following...Ch. 19 - Prob. 84PCh. 19 - Describe how naphthalene can he prepared from the...Ch. 19 - Prob. 86PCh. 19 - Using resonance contributors for the carbocation...Ch. 19 - Prob. 88PCh. 19 - When heated with chromic acid, compound A forms...Ch. 19 - Prob. 90PCh. 19 - Prob. 91PCh. 19 - What reagents are required to carry out the...Ch. 19 - Show how the following compounds can be prepared...Ch. 19 - An unknown compound reacts with ethyl chloride and...Ch. 19 - How can you distinguish the following compounds...Ch. 19 - P-Fluoronitrobenzene is more reactive toward...Ch. 19 - a. Explain why the following reaction leads to the...Ch. 19 - Describe how mescaline can be synthesized from...Ch. 19 - Propose a mechanism for the following reaction...Ch. 19 - Explain why hydroxide ion catalyzes the reaction...Ch. 19 - Propose a mechanism for each of the following...Ch. 19 - Prob. 102PCh. 19 - Prob. 103PCh. 19 - Describe how 3-methyl-1-phenyl-3-pentanol can he...Ch. 19 - a. How can aspirin be synthesized from benzene? b....Ch. 19 - Prob. 106PCh. 19 - Show how Novocain, a painkiller used frequently by...Ch. 19 - Prob. 108PCh. 19 - Saccharin, an artificial sweetener, is about 300...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Propose a mechanism for this isomerization.arrow_forwardA step in a synthesis of PGE1 (prostaglandin E1, alprostadil) is the reaction of a trisubstituted cyclohexene with bromine to form a bromolactone. Propose a mechanism for formation of this bromolactone and account for the observed stereochemistry of each substituent on the cyclohexane ring. Alprostadil is used as a temporary therapy for infants born with congenital heart defects that restrict pulmonary blood flow. It brings about dilation of the ductus arteriosus, which in turn increases blood flow in the lungs and blood oxygenation.arrow_forwardThe Meerwein-Ponndorf-Verley reaction involves reduction of a ketone by treatment with an excess of aluminum triisopropoxide, [(CH3)2CHO]3Al. The mechanism of the process is closely related to the Cannizzaro reaction in that a hydride ion acts as a leaving group. Propose a mechanism.arrow_forward
- Acid-catalyzed hydrolysis of the following epoxide gives a trans diol. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?arrow_forwardWhen (ft)-6-bromo-2,6-dimethylnonane is dissolved in CH3OH, nucleophilic substitution yields an optically inactive solution. When the isomeric halide (fl)-2-bromo-2,5- dimethylnonane is dissolved in CH3OH under the same conditions, nucleophilic substitution forms an optically active solution. Draw the products formed in each reaction, and explain why the difference in optical activity is observed.arrow_forwardShown below is the 1H NMR spectrum of the alkyl bromide used to make the phosphonium ylide that reacts with a ketone in a Wittig reaction to form a compound with molecular formula C11H14. What product is obtained from the Wittig reaction?arrow_forward
- Treatment of compound A (C8H17Br) with NaOCH2CH3 affords twoconstitutional isomers B and C. Ozonolysis of B affords CH2=O and(CH3CH2CH2)2C=O. Ozonolysis of C affords CH3CH2CH2COCH3 andCH3CH2CHO. What is the structure of A?arrow_forwardTreatment of compound A (C8H17Br) with NaOCH2CH3 affords two constitutional isomers B and C. Ozonolysis of B affords CH2=O and (CH3CH2CH2)2C=O. Ozonolysis of C affords CH3CH2CH2COCH3 and CH3CH2CHO. What is the structure of A?arrow_forwardShown below is the 1H NMR spectrum of the alkyl bromide used to make the phosphonium ylide that reacts with a ketone in a Wittig reaction to form acompound with molecular formula C11H14. What product is obtained from the Wittig reaction?arrow_forward
- The poison gelsemoxonine can be isolated from the leaves of a plant native to southeastern Asia (Gelsemium elegans). A key step in a synthesis of this natural product involves a thermally initiated (70°C) sigmatropic rearrangement of the compound shown below (R=protecting group) (J. Am. Chem. Soc. 2011, 133, 17634–17637). Draw the curved arrows for the mechanism of the formation of gelsemoxonine. Include lone pairs in your answer.arrow_forwardDraw and explain the mechanism in the reaction from compound 8, 9 to compound 10 e) NaH, RT, 72 h, 73%, >19:1 (E):(Z)arrow_forward(S)-3-methylpentan-2-one.(b) When cis-2,4-dimethylcyclohexanone is dissolved in aqueous ethanol containing atrace of NaOH, a mixture of cis and trans isomers results. Propose a mechanism forthis isomerization.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License