
Concept explainers
a)
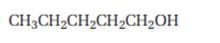
Interpretation:
Starting from 1-pentanol how pentanal can be prepared is to be shown.
Concept introduction:
Primary alcohols are oxidized to carboxylic acids by strong oxidizing agents like CrO3 and KMnO4 etc. The oxidation can be stopped in the
To show:
How to prepare pentanal starting from 1-pentanol.
b)
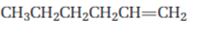
Interpretation:
Starting from 1-hexene how pentanal can be prepared is to be shown.
Concept introduction:
When treated with ozone,
To show:
How to prepare pentanal starting from 1-hexene.
c)
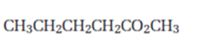
Interpretation:
Starting from methylpentanoate how pentanal can be prepared is to be shown.
Concept introduction:
Carboxylic acid derivatives like esters can be converted into aldehydes by reduction with diisobutylaluminium hydride (DIBAH) followed by acidification. The reaction is normally carried out at -78°C in toluene solution.
To show:
How to prepare pentanal starting from methylpentanoate.
d)
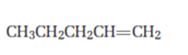
Interpretation:
Starting from 1-pentene how pentanal can be prepared is to be shown.
Concept introduction:
Alkenes when subjected to hydroboration-oxidation yield primary alcohols which when oxidized with Dess-Martin periodinate in dichloromethane yield aldehydes.
To show:
How to prepare pentanal starting from 1-pentene.
Trending nowThis is a popular solution!

Chapter 19 Solutions
ORGANIC CHEM.(LL)-W/OWL V2 >CUSTOM<
- Following is a retrosynthesis for the coronary vasodilator ganglefene. (a) Propose a synthesis for ganglefene from 4-hydroxybenzoic acid and 3-methyl-3-buten-2-one. (b) Is ganglefene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forwardWhat would be the major product if acetophenone (PhCOCH3) was treated with excess I2 and NaOH? A. CHCl3 B. PhCO2 C. PhCO2H D. CH3CO2 E. PhCH2OHarrow_forwardWhich of the following set of reagents will make cyclobutamine from bromocyclobutane? a NaN3 / ethanol and LiAlH4 in ether solution b NH3 and NaBH4 in ethanol solution c NaCN and LiAlH4 in ether solution d NH3 and H2 /Pdarrow_forward
- Which one of the following compounds gives 5-methyl-3-heptanol with LiAlH4 followed by water?arrow_forwardRank the compounds in each set in order of increasing acid strength.(a) CH3CH2COOH CH3CHBrCOOH CH3CBr2COOH(b) CH3CH2CH2CHBrCOOH CH3CH2CHBrCH2COOH CH3CHBrCH2CH2COOHarrow_forwardWhat would be the major product if diethyl malonate (EtO2CCH2CO2Et) was decarboxylated using H3O+ and heat? A. CH3CH2CO2H B. CH3CO2H C. CH2(CO2H)2 D. CH3CO2Etarrow_forward
- Treatment of compound A (C8H17Br) with NaOCH2CH3 affords two constitutional isomers B and C. Ozonolysis of B affords CH2=O and (CH3CH2CH2)2C=O. Ozonolysis of C affords CH3CH2CH2COCH3 and CH3CH2CHO. What is the structure of A?arrow_forwardhow to find the thiol that can be oxidized to produce propyl disulfide, CH3CH2CH2−S−S−CH2CH2CH3CH3CH2CH2−S−S−CH2CH2CH3.arrow_forwardWhat products would you expect from the oxidation of thefollowing compounds with (i) CrO3 in aqueous acid? (ii) withPCC?(a) tert-butanol(b) cyclohexanol(c) cyclohexanonearrow_forward
- (i) What is meant by chirality of a compound? Give an example.(ii) Which one of the following compounds is more easily hydrolyzed by KOH and why?CH3CHClCH2CH3 or CH3CH2CH2Clarrow_forwardPredict the products formed when CH3CH2¬C‚C≠- Na+ reacts with the following compounds.(a) ethyl bromide (b) tert-butyl bromide(c) formaldehyde (d) cyclohexanone(e) CH3CH2CH2CHOarrow_forwardDraw the product formed when phenylacetic acid (C6H5CH2COOH) istreated with following reagent. With some reagents, no reaction occurs. CH3NH2, DCCarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

