
MASTERINGCHEM FOR ORGANIC CHEM STANDAL
13th Edition
ISBN: 9781269517553
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19.6, Problem 13P
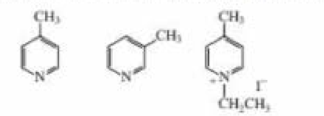
Rank the following compounds from easiest to hardest at removing a proton from its methyl substituent:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Rank the following compounds from easiest to hardest at removing a proton from its methyl substituent:
NCH3
N I + −CH3
CH2CH3
Please help me asap with the following question, I have a hard time getting it
Answer the following questions about each of the hydroxy ketones: 1-hydroxybutan-2-one (A) and 4-hydroxybutan-2-one (B).
a.) What is the molecular ion in the mass spectrum?b.) What IR absorptions are present in the functional group region?c.) How many lines are observed in the 13C NMR spectrum?d.) How many signals are observed in the 1H NMR spectrum?e.) Give the splitting observed for each type of proton as well as its approximate chemical shift.
Chapter 19 Solutions
MASTERINGCHEM FOR ORGANIC CHEM STANDAL
Ch. 19.1 - Name the following:Ch. 19.2 - Prob. 3PCh. 19.2 - Prob. 4PCh. 19.3 - Draw the product of each of the following...Ch. 19.5 - Prob. 6PCh. 19.5 - Explain why cyclopentadiene (pKa = 15) is more...Ch. 19.5 - When pyrrole is added to a dilute solution of...Ch. 19.6 - Prob. 10PCh. 19.6 - How to the mechanisms of the following reactions...Ch. 19.6 - Prob. 12P
Ch. 19.6 - Rank the following compounds from easiest to...Ch. 19.7 - Prob. 14PCh. 19.7 - Prob. 15PCh. 19.7 - Prob. 16PCh. 19.7 - Prob. 17PCh. 19.7 - Prob. 18PCh. 19.7 - Prob. 19PCh. 19.7 - Prob. 20PCh. 19 - Name the following:Ch. 19 - Prob. 22PCh. 19 - Rank the following compounds from strongest acid...Ch. 19 - Which of the following compounds is easier to...Ch. 19 - Rank the following compounds from most reactive to...Ch. 19 - One of the following compounds undergoes...Ch. 19 - Benzene undergoes electrophilic aromatic...Ch. 19 - Pyrrole reacts with excess...Ch. 19 - The dipole moments of furan and tetrahydrofuran...Ch. 19 - Name the following:Ch. 19 - Prob. 31PCh. 19 - Prob. 32PCh. 19 - a. Draw resonance contributors to show why...Ch. 19 - The chemical shifts of the C-2 hydrogen in the...Ch. 19 - Explain why protonating aniline has a dramatic...Ch. 19 - Prob. 36PCh. 19 - Propose a mechanism for the following reaction:Ch. 19 - Prob. 38PCh. 19 - Propose a mechanism for the following reactions:Ch. 19 - Prob. 40PCh. 19 - Prob. 41PCh. 19 - Prob. 42PCh. 19 - Organic chemists work with tetraphenylporphyrins...Ch. 19 - Show how the following compounds can be prepared...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Answer the following questions about each of the hydroxy ketones: 1hydroxybutan-2-one (A) and 4-hydroxybutan-2-one (B).a.What is the molecular ion in the mass spectrum? b. What IR absorptions are present in the functional group region? c. How many lines are observed in the 13C NMR spectrum? d.How many signals are observed in the 1H NMR spectrum? e.Give the splitting observed for each type of proton as well as its approximate chemical shift.arrow_forwardHow many non-equivalent protons are present in CH3CH═CH2?arrow_forwardWrite the expected product for each of the following reaction: 2-Butanone + NaCN, HCl -------------> ? Acetone + 2 eq. CH3CH2OH, H+ -----------> ? Benzaldehyde + 1 eq. benzyl alcohol, H+ ---------> ?arrow_forward
- Rank the compounds below in order of increasing acidity HF, H2O, NH3arrow_forwardCH3CH2CH2CH2Cl + 1) KCN 2) LiAlH4 3) H2O yields ___________. cyclopentylnitrile pentan-1-amine butan-1-nitrile pentan-1-nitrile 1-chloro-butan-1-nitrile Thank you!arrow_forwarddraw the major and minor resonance contributors and rank butanal, formaldehyde, and 2-pentanone from most to least electophylic. which is the least hydrated in h2o?arrow_forward
- Rank the following alcohols from strongest to weakest acid. b. Explain the relative acidities. CH2 = CHCH2OH CH3CH2CH2OH HC=CCH2OHarrow_forward1. Predict whether the compound below will test positive for each of the following qualitative tests: a) Tollens’ test, b) Benedict’s test 2. Provide at least one (1) test that could differentiate n-pentanol and pentanal and give the expected visible result/s. [n-pentanol and pentanal]arrow_forwardFor each compound, indicate the atom that is most apt to be protonated. a. CH3-CHOH-CH2NH2 b. CH3-C-NH2-CH3-OH c. CH3-C-CH3-NH2-CH2OHarrow_forward
- Will the bonding of the Zn2+ ion to the ketone oxygen lone pair make the ketone C=O easier or more difficult to reduce? Whyarrow_forwardDraw a mechanism of a reversible reaction of acetophenone and strong acid (H-A) to form a protonated acetophenone and a weak base. Draw all the electron pairs of the molecules and the arrows. Predict 1H NMR spectra of acetophenone and the protonated acetophenone.arrow_forwardDefine why are protons on an alpha carbon of an aldehyde or ketone more acidic than a proton on an alpha carbon of an ester? What are their pKa's?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY