
(a)
Interpretation:
The given substituted acetic acid has to be prepared using malonic ester synthesis.
Concept Introduction:
Malonic ester is a versatile starting material for the formation of new carbon-carbon bonds. It is because of the presence of acidic α-hydrogens between two carbonyl groups, the nucleophilicity of the enolate anion formed by the loss of α-hydrogen and the ability of the product to undergo decarboxylation after hydrolysis of ester.
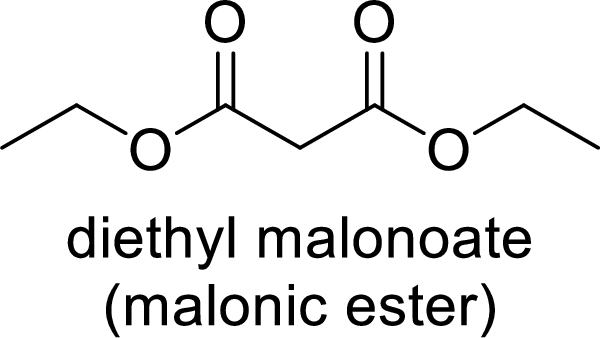
(b)
Interpretation:
The given substituted acetic acid has to be prepared using malonic ester synthesis.
Concept Introduction:
Malonic ester is a versatile starting material for the formation of new carbon-carbon bonds. It is because of the presence of acidic α-hydrogens between two carbonyl groups, the nucleophilicity of the enolate anion formed by the loss of α-hydrogen and the ability of the product to undergo decarboxylation after hydrolysis of ester.
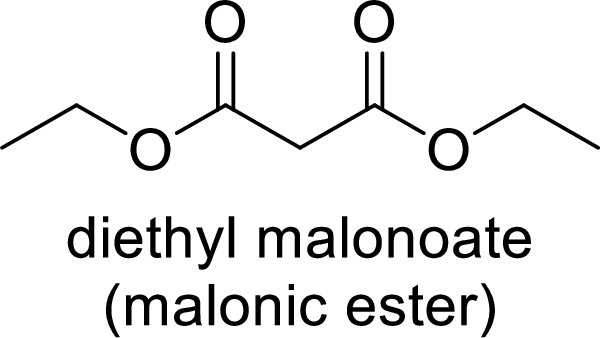
(c)
Interpretation:
The given substituted acetic acid has to be prepared using malonic ester synthesis.
Concept Introduction:
Malonic ester is a versatile starting material for the formation of new carbon-carbon bonds. It is because of the presence of acidic α-hydrogens between two carbonyl groups, the nucleophilicity of the enolate anion formed by the loss of α-hydrogen and the ability of the product to undergo decarboxylation after hydrolysis of ester.
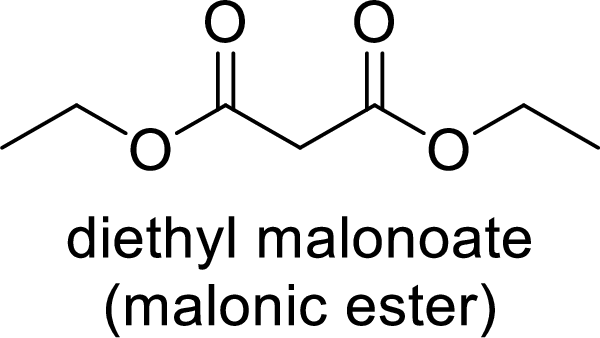
Trending nowThis is a popular solution!

Chapter 19 Solutions
OWLv2 with MindTap Reader, 1 term (6 months) Printed Access Card for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- The following molecule undergoes an intramolecular reaction in the presence of pyrrolidinium acetate, the protonated form of pyrrolidine. Draw the product of this reaction, assuming that a dehydration reaction takes place.arrow_forwardMalonic Ester Synthesis:arrow_forwardPrepare the following compounds starting from benzaldehyde and the appropriate ketone. Provide reactions for preparing the ketones starting from aromatic hydrocarbon compounds.arrow_forward
- Please give the appropriate reagents to complete the following synthesis.arrow_forwardPropose the synthesis for the following transformations.arrow_forwardDraw the structures of the initially formed enol tautomers in the reactions of propyne and dicyclohexylethyne with dicyclohexylborane followed by NaOHNaOH and H2O2H2O2arrow_forward
- complete the following synthesesarrow_forwardIsoamyl acetate (also known as isopentyl acetate) is an ester that is referred to as “banana flavor” due to its odor that resembles that of banana. It can be synthesized from isopentyl alcohol and acetic acid via nucleophilic acyl substitution mechanism. Propose a mechanism for the synthesis of isoamyl acetate from acetic acid and isoamyl alcohol.arrow_forwardSynthesize the product from the given material. Give the reagents necessary and draw out any intermediate products along the way.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
