
Concept explainers
(a)
Interpretation:
The preparation of the below given compound using directed aldol reactions has to be given.
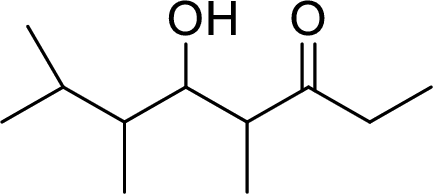
Concept Introduction:
In directed aldol reactions, the enolate anion is prepared with one carbonyl compound by LDA (Lithium diisopropylamide). The other carbonyl compound acts as an electrophile. Both of the carbonyl compounds have α-hydrogens, but only one enolate is prepared using LDA. When unsymmetrical ketone is used, it forms less substituted enolate.
LDA (Lithium diisopropylamide) is a strong base. It converts
(b)
Interpretation:
The preparation of the below given compound using directed aldol reactions has to be given.
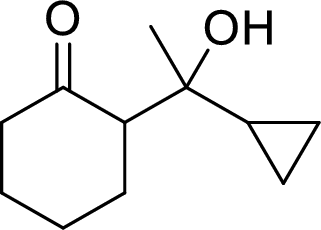
Concept Introduction:
In directed aldol reactions, the enolate anion is prepared with one carbonyl compound by LDA (Lithium diisopropylamide). The other carbonyl compound acts as an electrophile. Both of the carbonyl compounds have α-hydrogens, but only one enolate is prepared using LDA. When unsymmetrical ketone is used, it forms less substituted enolate.
LDA (Lithium diisopropylamide) is a strong base. It converts aldehydes, ketones and esters into their enolate anions.
Trending nowThis is a popular solution!

Chapter 19 Solutions
OWLv2 with MindTap Reader, 1 term (6 months) Printed Access Card for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- A student tried to prepare the following compounds using aldol condensations. Which of these compounds was she successful in synthesizing? Explain why the other syntheses were not successful.arrow_forwardPropose a crossed aldol reaction where the following compound would be the only product. Propose a mechanism for the reaction. OHarrow_forwardPropose an efficient synthesis for the following transformations using ONLY aldol, micheal and other reactions related to enol/enolate.arrow_forward
- What is the product formed when each dicarbonyl compound undergoes an intramolecular aldol reaction, followed by dehydration.arrow_forwardDraw the structure of the major aldol product (prior to possible dehydration) of the following reaction without specifying stereochemistry. 0 dilute aqueous NaOH 0-5° • You do not have to consider stereochemistry. • If no reaction occurs, draw the organic starting material. ChemDoodle 3barrow_forwardWhen the ketone and aldehyde shown below are treated with a base two possible products are formed. Discuss how each product is formed and provide a mechanism.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





