
Concept explainers
Mercury Emissions by Continent By weight, coal does not contain much mercury, but we burn a lot of it. Several industries besides coal-fired power plants contribute substantially to atmospheric mercury pollution. FIGURE 2.13 shows mercury emissions by industry from different regions of the world in 2010.
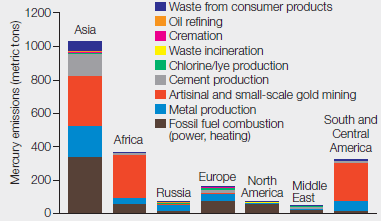
FIGURE 2.13 Global mercury emissions by sector, 2010.
About how many metric tons of mercury were released in total from these regions?
To determine: The total metric tons of mercury released from the regions given in the Fig. 2.13.
Introduction: Mercury is a toxic element and is naturally found in water, air, and soil. It is released in the environment through weathering of rocks, volcanic activity, and human activity. Human activity is mainly responsible for the mercury releases especially burning of coal in the coal-fired power stations, domestic uses, waste incinerators, industries, and mining of metals.
Explanation of Solution
As given in the problem statement, various industries besides coal-fired power plants contribute to mercury pollution in the atmosphere. Refer Fig. 2.13, “Global mercury emissions by sector, 2010” in the textbook. The graphical representation shows mercury emission (metric tons) by industries from different continents of the world in 2010. The data were reported from seven continents including Asia, Africa, Russia, Europe, North America, Middle East, and South and Central America. The mercury emissions released from various sectors included waste from consumer products, oil refining, cremation, waste incineration, mining, fossil fuel combustion, chlorine, cement, and metal production. On the basis of the data given in Fig. 2.13, Asia has the maximum mercury emissions reported as approximately 1100 metric tons. The approximate amount of mercury released by each continent is as follows:
- Asia – 1100 metric tons
- Africa – 380 metric tons
- South and Central America – 350 metric tons
- Europe – 180 metric tons
- North America – 80 metric tons
- Russia – 60 metric tons
- Middle East – 40 metric tons
By summing up the amount of mercury emissions from the seven continents, the total amount of mercury released (metric tons) comes to approximately 2200 metric tons.
The total mercury released by industry from different regions of the world in 2010 was 2200 metric tons.
Want to see more full solutions like this?
Chapter 2 Solutions
Biology: The Unity and Diversity of Life (Looseleaf)
Additional Science Textbook Solutions
MARINE BIOLOGY
Essentials of Genetics (9th Edition) - Standalone book
HUMAN ANATOMY
Campbell Biology: Concepts & Connections (8th Edition)
Genetic Analysis: An Integrated Approach (2nd Edition)
- Mercury Emissions by Continent By weight, coal does not contain much mercury, but we burn a lot of it. Several industries besides coal-fired power plants contribute substantially to atmospheric mercury pollution. FIGURE 2.13 shows mercury emissions by industry from different regions of the world in 2010. FIGURE 2.13 Global mercury emissions by sector, 2010. About how many metric tons of mercury were released from metal production in Asia?arrow_forwardMercury Emissions by Continent By weight, coal does not contain much mercury, but we burn a lot of it. Several industries besides coal-fired power plants contribute substantially to atmospheric mercury pollution. FIGURE 2.13 shows mercury emissions by industry from different regions of the world in 2010. FIGURE 2.13 Global mercury emissions by sector, 2010. Which industry tops the list of mercury emitters? Which industry is next on the list?arrow_forwardMercury Emissions by Continent By weight, coal does not contain much mercury, but we burn a lot of it. Several industries besides coal-fired power plants contribute substantially to atmospheric mercury pollution. FIGURE 2.13 shows mercury emissions by industry from different regions of the world in 2010. FIGURE 2.13 Global mercury emissions by sector, 2010. Which region emitted the most mercury from burning fossil fuels?arrow_forward
- Volcanoes emit much hywdrogen sulfide gas, H2S, which reacts with the oxygen in the air to form water and sulfur dioxide, SO2. Every 83 tons of H2S reacts with 117 tons of oxygen and forms 44 tons of water. How many tons SO2 are formed.arrow_forward38. The half-life of Potassium 40, on average, is 1,500 years 100,000 years 1.31 billion years 5730 years none of the abovearrow_forwardThe label on a candy bar says that it contains 150 Calories. If you could convert all of that energy to heat, you could raise the temperature of how much water by 15°C?arrow_forward
- The most abundant elements in the foods and beverage you consumables be oxygen,carbon, hydrogen,nitrogen. why might having these elements in consumables be useful?arrow_forwardWhy would increasing energy efficiency in homes and businesses across the united states reduce the bioaccumulation of mercury?arrow_forwardAll living things must be able to make proteins, and protein molecules always contain nitrogen. Explain how the nitrogen used for making proteins in a loin's body traveled from the atmosphere to the lion. How will it be returned to the atmosphere after the lion dies?arrow_forward
- Calculate the pH in water that is in equilibrium with calcite, but at the pre-industrial level of CO2 (280 ppm) and at the projected level in year 2100 with current international climate agreements (800 ppm). Ignore temperature and ionic strength effects.arrow_forwardThe smallest unit of an element that still retains thedistinctive behavior of that element is an ________.a. electronb. atomc. elemental particled. isotopearrow_forwardMercury Emissions by Continent By weight, coal does not contain much mercury, but we bum a lot of it. Several industries besides coal-fired power plants contribute substantially to atmospheric mercury pollution. FIGURE 2.12 shows mercury emissions by industry from different regions of the world in 2006. FIGURE 2.12 Global mercury emission, 2016. 2. Which industry tops the list of mercury emitters? Which industry is next on the list?arrow_forward
 Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781337408332Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781337408332Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning





