
Concept explainers
Figure 2.3 How many neutrons do carbon-12 and carbon-13 have, respectively?
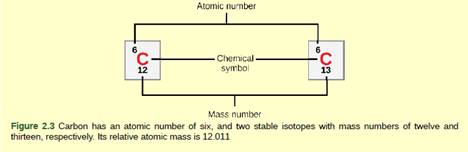
To write:
Number of neutrons in carbon-12 and carbon-13 respectively.
Introduction:
An atom consists of subatomic particles which are electrons, protons and neutrons. Every element in the periodic table has a different mass number and atomic number and based on that they have different number of subatomic particles. An atom consists of a nucleus which is composed of protons and neutrons. Neutrons as the name define; they are neutral in nature which means they don’t possess any electrical charge.
Explanation of Solution
The number of protons and neutrons in an atom of an element is called as its mass number and number of protons in an element is called as its atomic number. To calculate the number of neutrons, we need to subtract no of protons from the mass number.
Want to see more full solutions like this?
Chapter 2 Solutions
Biology 2e
Additional Science Textbook Solutions
College Physics
Microbiology with Diseases by Body System (5th Edition)
Biology: Life on Earth (11th Edition)
Human Biology: Concepts and Current Issues
Campbell Biology (11th Edition)
Microbiology: An Introduction
- Figure 2.3 How many neutrons do (K) potassium-39 and potassium-40 have, respectively?arrow_forwardPolonium is a rare element with 33 radioisotopes. The most common one, 210Po, has 82 protons and 128 neutrons. When 210Po decays, it emits an alpha particle, which is a helium nucleus (2 protons and 2 neutrons). 210Po decay is tricky to detect because alpha particles do not carry very much energy compared to other forms of radiation. For example, they can be stopped by a single sheet of paper or a few inches of air. That is one reason that authorities failed to discover toxic amounts of 210Po in the body of former KGB agent Alexander Litvinenko until after he died suddenly and mysteriously in 2006. What element does an atom of 210Po become after it emits an alpha particle?arrow_forwardAn isotope of sodium (Na) has a mass number of 22. How many neutrons does it have? a. 11 b. 12 c. 22 d. 44arrow_forward
- What frequency of EM radiation is emitted when an electron in a hydrogen atom jumps from n = 3 to n = 2? ( /2)Frequency = __________× 1014 Hzarrow_forwardCompared with 31P, the radioactive isotope 32P has(A) a different atomic number.(B) one more proton.(C) one more electron.(D) one more neutron.arrow_forwardHow X rays, gamma rays, and cosmic rays are more energetic than UV radiation ?arrow_forward
- Explain how the type of decay, the half life and the chemical properties make the following radionuclides suitable for the given applications: A single gram of polonium will reach a temperature of 500°C making it useful as a source of heat for space equipment (α emitter, half lives of polonium isotopes range from less than a second to 200 years).arrow_forwardRadioactivity is a phenomenon of the spontaneous emission of: A. protons (alpha particles) B. electrons (beta particles) C. gamma rays (short wave electromagnetic waves) D. All of the abovearrow_forwardAn isotope of sodium (Na) has a mass number of 22 and an atomic number of 11.how many neutrons does it have? 11 12 22 44arrow_forward
- a 139 ng sample of radioactive material has a half life of 4.2 days how many ng of radioactive material is left after 3 half lives ?arrow_forwardThe reason why a nucleus might emit radiation is because by doing so, it forms a more stable-lower energy nucleus.True falsearrow_forwardWhat is the unknown isotope in the following fission reaction: n + 235U → 131I + ? + 3nA. 86Rb B. 102Rb C. 89Y D. 102Yarrow_forward
 Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning





