
Microbiology with Diseases by Body System (5th Edition)
5th Edition
ISBN: 9780134477206
Author: Robert W. Bauman Ph.D.
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 1VI
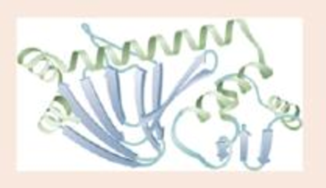
Label a portion of the molecule below; label two types of secondary structure; circle the tertiary structure.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Explain Four Levels of Protein Structure with examples and talk about the importance of each structure (Primary, Secondary, Tertiary, and Quaternary structure). With photos please
fill the following table
polymer or large biological molecule
monomer or smaller subunit
one funtion
name od covalent bond
nulceic acids
three fatty acids
easter bond
acts as an enzyme
immediate or long-term energy source
glycosidic linkage
Identify if the molecule below is a protein, lipid, nucleic acid or carbohydrate.
Chapter 2 Solutions
Microbiology with Diseases by Body System (5th Edition)
Ch. 2 - Electrons zip around the nucleus at about 5...Ch. 2 - Chlorine and potassium atoms form ionic bonds,...Ch. 2 - Why are decomposition reactions exothermic, that...Ch. 2 - Why does the neutralization of an acid by a base...Ch. 2 - Raw Oysters and Antacids: A Deadly Mix? The highly...Ch. 2 - Why do the cell membranes of microbes living in...Ch. 2 - Prob. 1MCCh. 2 - The atomic mass of an atom most closely...Ch. 2 - One isotope of iodine differs from another in...Ch. 2 - Prob. 4MC
Ch. 2 - Which of the following terms most correctly...Ch. 2 - In water, cations and anions of salts dissociate...Ch. 2 - Prob. 7MCCh. 2 - Which of the following statements about a...Ch. 2 - Proteins are polymers of ___________. a. amino...Ch. 2 - Which of the following are hydrophobic organic...Ch. 2 - Fill in the Blanks 1. The outermost electron shell...Ch. 2 - Fill in the Blanks 2. The type of chemical bond...Ch. 2 - Prob. 3FIBCh. 2 - Prob. 4FIBCh. 2 - Fill in the Blanks 5. Groups of atoms such as NH2...Ch. 2 - Fill in the Blanks 6. The reverse of dehydration...Ch. 2 - Fill in the Blanks 7. Reactions that release...Ch. 2 - Fill in the Blanks 8. All chemical reactions begin...Ch. 2 - Fill in the Blanks 9. The ____________ scale is a...Ch. 2 - Prob. 10FIBCh. 2 - Label a portion of the molecule below; label two...Ch. 2 - Shown is the amino acid tryptophan. Put the letter...Ch. 2 - List three main types of chemical bonds, and give...Ch. 2 - Name five properties of water that are vital to...Ch. 2 - Prob. 3SACh. 2 - What is the difference between atomic oxygen and...Ch. 2 - Explain how the polarity of water molecules makes...Ch. 2 - Prob. 1CTCh. 2 - Prob. 2CTCh. 2 - Two freshmen disagree about an aspect of...Ch. 2 - When an egg white is heated, it changes from...Ch. 2 - Prob. 5CTCh. 2 - The poison glands of many bees and wasps contain...Ch. 2 - Prob. 7CTCh. 2 - Prob. 8CTCh. 2 - The deadly poison hydrogen cyanide has the...Ch. 2 - Triple covalent bonds are stronger and more...Ch. 2 - How can hydrogen bonding between water molecules...Ch. 2 - How can a single molecule of magnesium hydroxide...Ch. 2 - Prob. 13CTCh. 2 - Prob. 14CTCh. 2 - A textbook states that only five nucleotide bases...Ch. 2 - Using the following terms, fill in the following...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Choose from the columns the compound that correctly represents the functional groups listed (rows). Structure of compounds are shown below.arrow_forwardLabel all structures as appropriate. Draw and label all appropriate molecules to complet the pictures.arrow_forwardPlease help me identify the correct level each image represents. Answer Choices are: Secondary Structure Quaternary Structure Primary Structure Tertiary Structurearrow_forward
- Match the molecule with its correct descriptionarrow_forwardExplain Four Levels of Protein Structure with examples and talk about the importance of each structure (Primary, Secondary, Tertiary, and Quaternary structure). Please write long answers for each structure.arrow_forwardDefine the following terms: a. carbonyl group b. carboxyl group c. amino group d. hydroxyl group e. peptide bondarrow_forward
- list the elements, monomer name & basic shape, common example, structures formed and functions in organisms, other unique features, the positive and negative colors of nucleic acidarrow_forwardDescribe the structure, mention the alpha helices, beta sheets, and anything else that the picture of the structure has.arrow_forwardWhich of the following types of bonds are present in the primary structures of proteins? Covalent bonds, hydrogen bonds and dislufide linkages Ionic interactions Hydrogen bonds and covalent bonds Covalent bondarrow_forward
- The type of molecule in the above question is the MONOMER of which of the following polymers? Lipids Polysaccharides Nucleic acids Proteinsarrow_forwardlist the elements, monomer name & basic shape, common example, structures formed and functions in organisms, other unique features, the positive and negative colors of proteinarrow_forwardPlease explain compounds and chemical bonds, ionic, covalent, and hydrogen bondsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning

Biology Today and Tomorrow without Physiology (Mi...
Biology
ISBN:9781305117396
Author:Cecie Starr, Christine Evers, Lisa Starr
Publisher:Cengage Learning
Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license