
Concept explainers
(a)
Interpretation: The conjugate base of given acid is to be stated.
Concept introduction: An acid is a substance that is capable to donate a
Answer to Problem 2.36P
The conjugate base of given acid is
Explanation of Solution
The given acid is
An acid is a substance that is capable to donate a
Thus, the
Hence, the conjugate base of given acid is
The conjugate base of given acid is
(b)
Interpretation: The conjugate base of given acid is to be stated.
Concept introduction: An acid is a substance that is capable to donate a
Answer to Problem 2.36P
The conjugate base of given acid is
Explanation of Solution
The given acid is
An acid is a substance that is capable to donate a
Thus, the
Hence, the conjugate base of given acid is
The conjugate base of given acid is
(c)
Interpretation: The conjugate base of given acid is to be stated.
Concept introduction: An acid is a substance that is capable to donate a
Answer to Problem 2.36P
The conjugate base of given acid is
Explanation of Solution
The given acid is
An acid is a substance that is capable to donate a
Thus, the
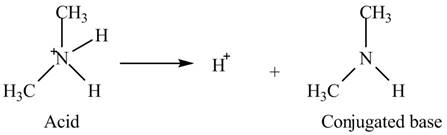
Figure 1
Hence, the conjugate base of given acid is
The conjugate base of given acid is
(d)
Interpretation: The conjugate base of given acid is to be stated.
Concept introduction: An acid is a substance that is capable to donate an
Answer to Problem 2.36P
The conjugate base of given acid is
Explanation of Solution
The given acid is
An acid is a substance that is capable to donate a
Thus, the
Hence, the conjugate base of given acid is
The conjugate base of given acid is
(e)
Interpretation: The conjugate base of given acid is to be stated.
Concept introduction: An acid is a substance that is capable to donate a
Answer to Problem 2.36P
The conjugate base of given acid ispropionate ion.
Explanation of Solution
The given acid is propionic acid.
An acid is a substance that is capable to donate a
Thus, the
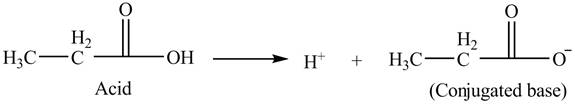
Figure 2
Hence, the conjugate base of given acid is propionate.
The conjugate base of given acid is shown in figure 2.
(f)
Interpretation: The conjugate base of given acid is to be stated.
Concept introduction: An acid is a substance that is capable to donate a
Answer to Problem 2.36P
The conjugate base of given acid is
Explanation of Solution
The given acid is
The given acid is ethynylcyclohexane.
An acid is a substance that is capable to donate a
Thus, the
Hence, the conjugate base of given acid is
The conjugate base of given acid is
Want to see more full solutions like this?
Chapter 2 Solutions
Connect Access Card For Organic Chemistry
- Which of the following do not represent a conjugate acid-base pair? For those pairs that are not conjugate acid-base pairs, write the correct conjugate acid-base pair for each species in the pair. a. HI, I- b. HClO, HClO2 c. H3PO4, PO43- d. H2CO3, CO32-arrow_forward. Choose pairs in which the species listed first is the conjugate base of the species listed second. a. S2-, HS- b. H+, OH- c. HBr, Br- d. NO2, HNOarrow_forwardWrite equations that show NH3 as both a conjugate acid and a conjugate base.arrow_forward
- Consider the following four solutions: (1) apple juice, pH 3.8, (2) pickle juice, pH 3.5, (3) carbonated beverage, pH 3.0, and (4) drinking water, pH 7.2. a. Which solution has the highest [H3O+]? b. Which solution has the highest [OH]? c. List the solutions in order of increasing acidity. d. List the solutions in order of decreasing basicity.arrow_forwardHow is acid strength related to the value of Ka? What is the difference between strong acids and weak acids (see Table 13-1)? As the strength of an acid increases, what happens to the strength of the conjugate base? How is base strength related to the value of Kb? As the strength of a base increases, what happens to the strength of the conjugate acid?arrow_forwardThe conjugate base of hydrofluoric acid dissolved in water is: a F b OH c H3O d HF e F2arrow_forward
- Consider the following four biological solutions: (1) bile, pH 8.0, (2) blood, pH 7.4, (3) urine, pH 6.0, and (4) gastric juice, pH 1.6. a. Which solution has the lowest [H3O+]? b. Which solution has the lowest [OH]? c. List the solutions in order of decreasing acidity. d. List the solutions in order of increasing basicity.arrow_forward12.63 For each of the following reactions, indicate the Bronsted-Lowrv acids and bases. What are the conjugate acid-base pairs? CN’(aq) + H2O(€) «=* HCN(aq) + OH’(aq) HCO}-(aq) + H,o+(aq) +* H2CO,(aq) + H,O(€) (C) CH,CtX)H(aq) + HS~(aq)i=i CH}COO"(aq) + H2S(aq)arrow_forwardWhat is the conjugate acid of each of the following? What is the conjugate base of each?. (a) OH-. (b) H2O. (c) HCO3-. (d) NH3. (e) HSO4-. (f) H2O2. (g) HS-. (h) H5N2+arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning





