
Concept explainers
(a)
Interpretation:
Given uncharged molecule is to be identified as polar or nonpolar, using its electrostatic potential map. If the molecule is polar, the direction of its net molecular dipole moment it to be determined.
Concept introduction:
A polar bond is one in which the bond pair is unequally shared by the two atoms.
A partial positive charge is developed on the less electronegative atom while and an equal but negative partial charge is developed on the more electronegative atom.
Molecules that contain more than one polar bond may or may not have a net dipole moment. The dipole moment is a vector quantity. The net molecular dipole moment is the result of the vector addition of all the individual dipole moments. Depending on the symmetry of the molecule, the individual bond dipoles can partly or completely cancel or reinforce each other.
A bond dipole or a molecular dipole is represented by an arrow pointing from the atom or region with a partial positive charge toward an atom or region with a partial negative charge.
Electrostatic potential maps of molecules show the distribution of electron density in different parts of the molecule. The electron density is represented by different colors, ranging from blue to red. Blue color indicates a low electron density, an atom or region with a partial positive charge. Red color indicates high electron density, an atom or region with a partial negative charge.
Answer to Problem 2.40P
The electrostatic potential map shows that the molecule is nonpolar.
Explanation of Solution

The electrostatic potential map shows the molecule with a negative charge concentrated at the center, with positive charge distributed symmetrically around the center. This shows that the individual bond dipoles are all of equal magnitude, and they all point toward the center. The vector addition of these dipoles will be zero because of their symmetric distribution. Therefore, the molecule is nonpolar.
The net dipole moment of a molecule is the vector sum of the individual bond dipoles.
(b)
Interpretation:
Given uncharged molecule is to be identified as polar or nonpolar, using its electrostatic potential map. If the molecule is polar, the direction of its net molecular dipole moment it to be determined.
Concept introduction:
A polar bond is one in which the bond pair is unequally shared by the two atoms.
A partial positive charge is developed on the less electronegative atom while and an equal but negative partial charge is developed on the more electronegative atom.
Molecules that contain more than one polar bond may or may not have a net dipole moment. The dipole moment is a vector quantity. The net molecular dipole moment is the result of the vector addition of all the individual dipole moments. Depending on the symmetry of the molecule, the individual bond dipoles can partly or completely cancel or reinforce each other.
A bond dipole or a molecular dipole is represented by an arrow pointing from the atom or region with a partial positive charge toward an atom or region with a partial negative charge.
Electrostatic potential maps of molecules show the distribution of electron density in different parts of the molecule. The electron density is represented by different colors, ranging from blue to red. Blue color indicates a low electron density, an atom or region with a partial positive charge. Red color indicates high electron density, an atom or region with a partial negative charge.
Answer to Problem 2.40P
The electrostatic potential map shows that the molecule is nonpolar.
Explanation of Solution
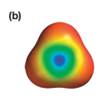
The electrostatic potential map shows a molecule with a negative charge concentrated at the center, with positive charge distributed symmetrically around the center. This shows that the individual bond dipoles are all of equal magnitude, and they all point toward the center. The vector addition of these dipoles will be zero because of their symmetric distribution. Therefore, the molecule is nonpolar.
The net dipole moment of a molecule is the vector sum of the individual bond dipoles.
(c)
Interpretation:
The given uncharged molecule is to be identified as polar or nonpolar, using its electrostatic potential map. If the molecule is polar, the direction of its net molecular dipole moment it to be determined.
Concept introduction:
A polar bond is one in which the bond pair is unequally shared by the two atoms.
A partial positive charge is developed on the less electronegative atom while and an equal but negative partial charge is developed on the more electronegative atom.
Molecules that contain more than one polar bond may or may not have a net dipole moment. The dipole moment is a vector quantity. The net molecular dipole moment is the result of the vector addition of all the individual dipole moments. Depending on the symmetry of the molecule, the individual bond dipoles can partly or completely cancel or reinforce each other.
A bond dipole or a molecular dipole is represented by an arrow pointing from the atom or region with a partial positive charge toward an atom or region with a partial negative charge.
Electrostatic potential maps of molecules show the distribution of electron density in different parts of the molecule. The electron density is represented by different colors, ranging from blue to red. Blue color indicates a low electron density, an atom or region with a partial positive charge. Red color indicates high electron density, an atom or region with a partial negative charge.
Answer to Problem 2.40P
The electrostatic potential map shows that the molecule is polar.
The direction of the net molecular dipole is downward as shown below.
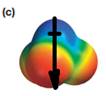
Explanation of Solution
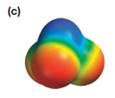
The electrostatic potential map shows a molecule with an asymmetric charge distribution. The positive charge is concentrated on the atom at the top, while the negative charge is distributed on three atoms at the bottom. The individual bond dipoles will therefore not cancel completely. Therefore, the molecule is polar.
The direction of the molecular dipole will be downward, as shown below by the black arrow.
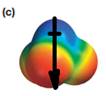
The net dipole moment of a molecule is the vector sum of the individual bond dipoles.
(d)
Interpretation:
The given uncharged molecule is to be identified as polar or nonpolar, using its electrostatic potential map. If the molecule is polar, the direction of its net molecular dipole moment it to be determined.
Concept introduction:
A polar bond is one in which the bond pair is unequally shared by the two atoms.
A partial positive charge is developed on the less electronegative atom while and an equal but negative partial charge is developed on the more electronegative atom.
Molecules that contain more than one polar bond may or may not have a net dipole moment. The dipole moment is a vector quantity. The net molecular dipole moment is the result of the vector addition of all the individual dipole moments. Depending on the symmetry of the molecule, the individual bond dipoles can partly or completely cancel or reinforce each other.
A bond dipole or a molecular dipole is represented by an arrow pointing from the atom or region with a partial positive charge toward an atom or region with a partial negative charge.
Electrostatic potential maps of molecules show the distribution of electron density in different parts of the molecule. The electron density is represented by different colors, ranging from blue to red. Blue color indicates a low electron density, an atom or region with a partial positive charge. Red color indicates high electron density, an atom or region with a partial negative charge.
Answer to Problem 2.40P
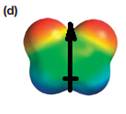
The electrostatic potential map shows that the molecule is polar.
The direction of the net molecular dipole is upward, as shown below.
Explanation of Solution
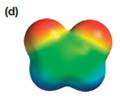
The electrostatic potential map shows a molecule with a negative charge distributed on two atoms at the top and the positive charge distributed on two atoms at the bottom. This shows that the individual bond dipoles both point approximately upward and slightly away from the center line. The vector addition of these dipoles will be nonzero. Therefore, the molecule is polar.
The direction of the net dipole moment will be upward as shown below.
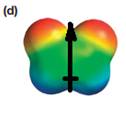
The net dipole moment of a molecule is the vector sum of the individual bond dipoles.
(e)
Interpretation:
The given uncharged molecule is to be identified as polar or nonpolar, using its electrostatic potential map. If the molecule is polar, the direction of its net molecular dipole moment it to be determined.
Concept introduction:
A polar bond is one in which the bond pair is unequally shared by the two atoms.
A partial positive charge is developed on the less electronegative atom while and an equal but negative partial charge is developed on the more electronegative atom.
Molecules that contain more than one polar bond may or may not have a net dipole moment. The dipole moment is a vector quantity. The net molecular dipole moment is the result of the vector addition of all the individual dipole moments. Depending on the symmetry of the molecule, the individual bond dipoles can partly or completely cancel or reinforce each other.
A bond dipole or a molecular dipole is represented by an arrow pointing from the atom or region with a partial positive charge toward an atom or region with a partial negative charge.
Electrostatic potential maps of molecules show the distribution of electron density in different parts of the molecule. The electron density is represented by different colors, ranging from blue to red. Blue color indicates a low electron density, an atom or region with a partial positive charge. Red color indicates high electron density, an atom or region with a partial negative charge.
Answer to Problem 2.40P
The electrostatic potential map shows that the molecule is polar.
The direction of the net molecular dipole is upward as shown below.
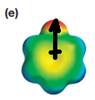
Explanation of Solution
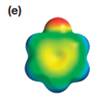
The electrostatic potential map shows a molecule with a negative charge concentrated on the atom at the top center, with positive charge distributed over atoms on the side and at the bottom. This shows that the individual bond dipoles are not symmetrically distributed and will not cancel out completely. Therefore, the molecule is polar.
The direction of the net molecular dipole will be upward because of the concentration of negative charge at the top and a symmetrical distribution of the positive charge in the rest of the molecule.
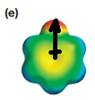
The net dipole moment of a molecule is the vector sum of the individual bond dipoles.
(f)
Interpretation:
The given uncharged molecule is to be identified as polar or nonpolar, using its electrostatic potential map. If the molecule is polar, the direction of its net molecular dipole moment it to be determined.
Concept introduction:
A polar bond is one in which the bond pair is unequally shared by the two atoms.
A partial positive charge is developed on the less electronegative atom while and an equal but negative partial charge is developed on the more electronegative atom.
Molecules that contain more than one polar bond may or may not have a net dipole moment. The dipole moment is a vector quantity. The net molecular dipole moment is the result of the vector addition of all the individual dipole moments. Depending on the symmetry of the molecule, the individual bond dipoles can partly or completely cancel or reinforce each other.
A bond dipole or a molecular dipole is represented by an arrow pointing from the atom or region with a partial positive charge toward an atom or region with a partial negative charge.
Electrostatic potential maps of molecules show the distribution of electron density in different parts of the molecule. The electron density is represented by different colors, ranging from blue to red. Blue color indicates a low electron density, an atom or region with a partial positive charge. Red color indicates high electron density, an atom or region with a partial negative charge.
Answer to Problem 2.40P
The electrostatic potential map shows that the molecule is nonpolar.
Explanation of Solution
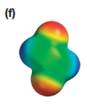
The electrostatic potential map shows a charge distribution that is symmetric about the center of the molecule with two negative regions opposite each other across the center as well as two positive regions across the center. The individual bond dipoles will therefore cancel out completely. Therefore, the net dipole moment will be zero, and the molecule will be nonpolar.
The net dipole moment of a molecule is the vector sum of the individual bond dipoles.
Want to see more full solutions like this?
Chapter 2 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- 3. Which of the following molecules are conjugated? For those which are conjugated, write a resonance structure.arrow_forwardA. Classify the following molecules as polar or non-polar (write below the chemical formula in the first column. Explain your answer in terms of the (a) Lewis structure (b) molecular geometry and (c) bond polarity. (You can add rows if you need more spaces for your explanation.) Hint: Although I am not asking you to give the no. of bonding and non-bonding pairs, you have to do it to be able to identify the electron pair geometry and molecular geometry. Note: Items #1 to #4 are hypothetical molecules. The number of valence electron/s of the hypothetical atoms are given in parenthesis.) There is no need to give actual molecules to represent them. Polar or Non-polar Explanation 1. Z2 (Z =7)arrow_forwardUne or more valid answers. Explain the answers. The localization or resonance energy: a) Justify the increased stability of molecules that have unlocated electrons b) It is a consequence of the mobility of the electrons that form the “sigma” bonds c) It occurs in compounds with conjugated double bonds, but not in aromatic compounds d) It is shown by the experimental values of the enthalpies of formation.arrow_forward
- A. Classify the following hypothetical molecules as polar or non-polar. Note: Number 5 item is not a hypothetical molecule. (The number of valence electrons are given in parenthesis.) Explain your answer in terms of: (a) Lewis structure (b) Molecular geometry (c) Symmetry (symmetrical or asymmetrical) (d) Dipole moment (descriptive) Molecule/ Ion (No. of Valence Non-polar electron) 2. HG3 (H=7, G=7) Polar or Explanationarrow_forwardConstruct, fully label, and populate a MO diagram for the nitrosonium anion, NO+. Based on the diagram, state which of NO+ or NO you expect to have a stronger bond.arrow_forwardInstructions: Complete the table. For the % s-character, type in only the number, with no percentage sign (e.g. 15)For the bond angle, type in only the number, with no degrees sign (e.g. 60)For the common name, type in using small letters only.For the molecular formula and degree of hybridization, type in the symbol and number together (e.g. CCl3)arrow_forward
- True or False: In resonance energy transfer, the excited electron decays down to an orbital in the same molecule. (a). True (b).Falsearrow_forwardBF3 is a molecule with ["3", "2", "1", "4"] electron domains giving it a ["linear", "trigonal planar", "tetrahedral"] electron domain geometry. It has ["3", "4", "2", "1"] bonded atoms and ["1", "2", "0", "3"] lone pairs. BF3 will exhibit a ["linear", "trigonal planar", "trigonal pyramidal", "tetrahedral", "bent"] molecular geometry.arrow_forwardGiven the following two negatively charged molecules. Draw in the pi orbitals. In which of the two molecules is the negative charge the most stabilized. WHY???arrow_forward
- (a) In one of the two boxes below, draw a wedge and dashed wedge structure (picture) of CH3Cl that best illustrates the geometry about the central atom. In the other box, draw another picture of the model from a different angle (viewpoint). (b) In CH3Cl, are the three hydrogen atoms equivalent (i.e., do they have identical environments with respect to the other atoms adjacent to themselves)? Briefly explain the evidence for your answerarrow_forwardThank you! which one contributes most and which least to the real structure of the cyanate ion?arrow_forwardNitromethane is an organic compound with the molecular Draw a complete Lewis structure for the conjugate acid of nitromethane that shows all bonds, unshared electron pairs, and minimized formal charges, where appropriate. formula CH, NO,. Draw a complete Lewis structure that shows all bonds, unshared electron pairs, and minimized formal charges, where appropriate. H H 1. H : 0 :arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
