
Concept explainers
Interpretation:
The reason should be explained for the third period in the periodic table contains eight elements.
Concept introduction:
The Aufbau principle:
The aufbau principle is states that in the ground state of an atom, electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels. For example, the 1s shell is filled before the 2s subshell is occupied.
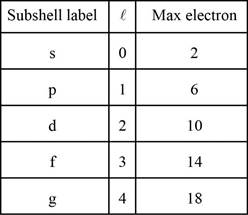
The subshell ordering by this rule is
Number of electrons in the shells is given below,
Each shell contains only a fixed number of electrons. The first shell contains two electrons, the second shell contains eight (2 + 6) electrons, the third shell contains 18 (2 + 6 + 10), fourth shell contains (2 + 6 + 10 + 14 ) and so on.
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
FUND. GEN. ORG&BIO CHEM PKG
- what makes Isotopes of an element different from one another?arrow_forwardHow does the atomic structure or composition of the versions of sodium in question C above differ from a typical sodium atom, with its atomic mass of 23?arrow_forwardwhy do the sizes of the transition elements change more gradually than those of the representative elements?arrow_forward
- Which electron shell is the valence shell of an atom, and what is its significance?arrow_forwardAnother major use of bismuth has been as an ingredient in low-melting metal alloys, such as those used in firesprinkler systems and in typesetting. The element itself is a brittle white crystalline solid. How do these characteristicsfit with the fact that bismuth is in the same periodic group with such nonmetallic elements as nitrogenand phosphorus?arrow_forwardWhich element has only one proton?arrow_forward
- If one compound has the formula C5H10 and another has the formula C4H10, are the two compounds isomers? Explain.arrow_forwardA principal shell with a value of n=3 would contain s,p,d, and f orbitals true or falsearrow_forwardIn the periodic table, the elements are arrangedaccording to ________ .a. size c. mass numberb. charge d. atomic numberarrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning


